TOPIC 3 CHEMICAL BONDING, FORMULA AND NOMENCLATURE – CHEMISTRY FORM TWO
Introduction
Chemical bonding involves holding atoms together to form molecules or compounds. A chemical formula refers to symbols and numbers that represent the composition of a particular chemical substance. Nomenclature means naming.
In this chapter, you will learn about the concept of chemical bonding, the concept of chemical formulas, the determination of empirical and molecular formulas of common compounds and the nomenclature of binary inorganic compounds using the IUPAC system. The competencies developed will enable you to explore the relationships between chemical substances and apply the acquired knowledge in understanding properties of different materials used in daily life.
Think: Contribution of chemical bonding, formulas, and nomenclature to global processes and activities
Concept of chemical bonding
Task 1
Use videos, interactive simulations, or other reliable resources to explore the concept of chemical bonding.
A bond is anything that holds two or more substances together. Many things used in our daily lives are constructed using different materials joined together by some bonds. For example, in a brick wall (Figure 3.1), each brick is joined to the other by a bond made of mortar. Similarly, chemical substances are made of atoms that are held together by chemical bonds.

Bricks bonded using mortar to form a wall
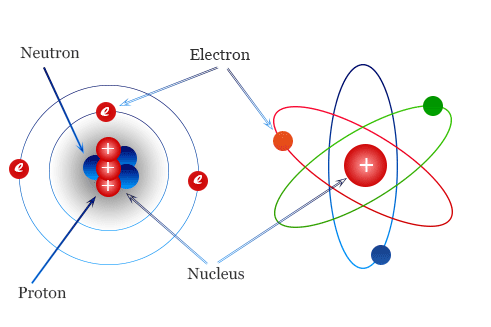
A chemical bond is a force of attraction that holds atoms or ions together to form molecules or compounds. The bond may result from forces of attraction between oppositely charged ions or through the sharing of electrons.
Note: A molecule is the smallest particle of an element or compound which can normally exist separately.
CHEMICAL BONDING
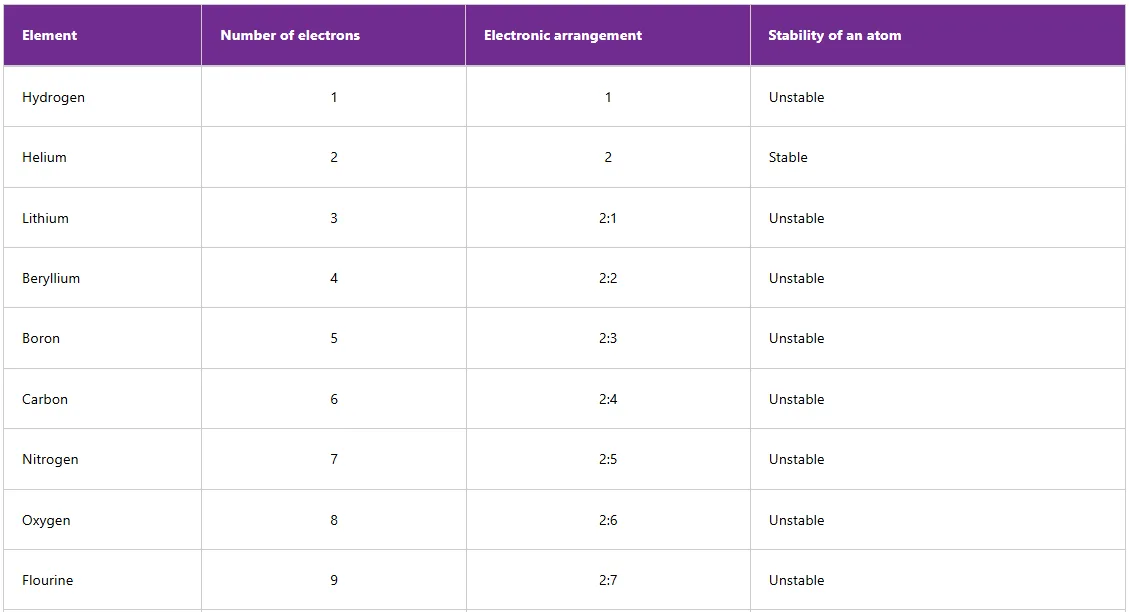
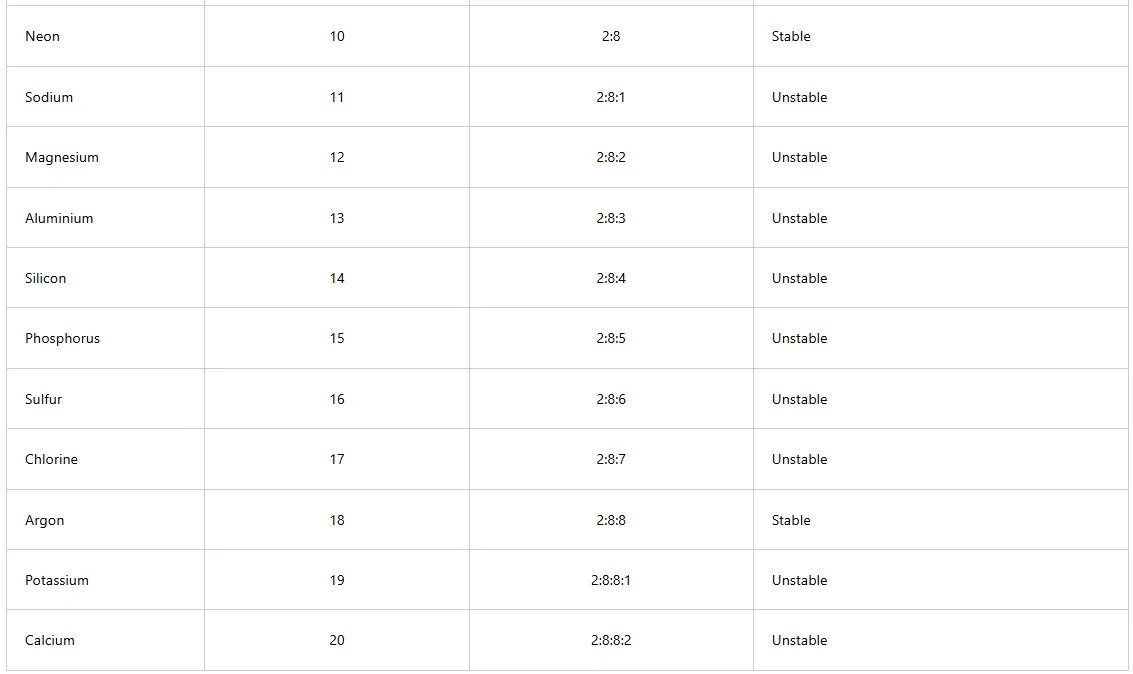
Chemical bonding involves electrons in the outermost shells of atoms. When the outermost shells are completely filled with electrons, the atoms are said to be stable, otherwise they are unstable. Table 3.1 shows the electronic arrangements of some elements and the stability of atoms of the elements.
From Table below, only helium, neon, and argon have stable electronic arrangements. Helium has a maximum number of 2 electrons in its outermost shell. Neon and argon have a maximum of 8 electrons in each of their respective outermost shells. These stable atoms are generally unreactive and can exist freely as single atoms. Unstable atoms cannot exist freely as single atoms. For unstable atoms to become stable, they should acquire electronic arrangements similar to those of noble gases. This means that they can either lose, gain or share electrons through chemical bonding.
Table 1: Electronic arrangements and stability of the first twenty elements of the periodic table
FORMATION OF IONS
When an atom loses an electron to attain a stable electronic arrangement, the lost electron is transferred to another atom making it stable as well. The resulting species become ions. The one that loses an electron becomes a positively charged ion, while the one that gains an electron becomes a negatively charged ion.
The number of electrons gained or lost will be equal to the charge of an ion. The positively charged ion is called a cation and the negatively charged ion is called an anion. Electrons are negatively charged while protons are positively charged; thus, the charge of the ion is due to the unbalanced number of electrons and protons.
For example, a sodium atom has eleven protons and eleven electrons. Its charge is 0 because (+11) + (–11) = 0. After sodium loses one electron it will have 10 electrons. Its charge will become +1 because (+11) + (–10) = +1 (Figure below).

Electron arrangement in sodium atom and its ion
ELECTROVALENT BONDING
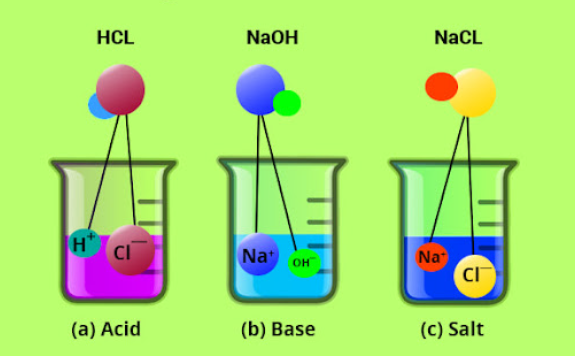
Electrovalent bonding, also known as ionic bonding, is a type of chemical bond formed when one atom transfers electrons to another, resulting in the formation of positively and negatively charged ions. These charged ions attract each other due to their opposite charges.
Electrovalent bonding usually occurs between a metal and a non-metal. The metal loses electron(s) and the non-metal gains electron(s). For example, when sodium and chlorine react to form sodium chloride, the sodium atom must lose an electron to acquire a stable noble gas structure of neon, while the chlorine atom must gain an electron to acquire a stable noble gas structure of argon.
This results in a positively charged sodium ion and a negatively charged chloride ion which attract each other to form sodium chloride as shown in Figure 3.3. For this chapter, electrons are represented by dots with different colours.

Electrovalent bonding in sodium chloride
Molecules of electrovalent compounds are not discrete. This means that the electrons are not localised or attached to particular ions of opposite charges to form pairs. Instead, a group of cations surrounds an anion and a group of anions surround a cation. This means that ions can move freely around each other, especially when in molten or in solution forms.
Properties of electrovalent compounds
Electrovalent (ionic) compounds have the following properties:
(a) They are generally soluble in water.
(b) They conduct electricity in solution or molten forms but not in their solid forms.
(c) They are usually crystalline solids at room temperature.
(d) They have high melting and boiling points.
(e) They are generally insoluble in non-polar solvents such as carbon tetrachloride and hexane. Non-polar solvents contain bonds between atoms with similar electronegativities.
(f) They are typically hard but brittle.
(g) They have high densities.
Activity 1
Aim: To investigate the electrovalent bonding in substances
Requirements: Table salt (sodium chloride), cooking oil, slaked lime, baking powder, sugar, three glass beakers, distilled water, a tablespoon, an electrical conductivity meter (or a small LED bulb with wires and a battery), and three stirring rods
Procedure
1. Fill a glass beaker with distilled water and add 2 tablespoons of table salt (sodium chloride). Stir with the stirring rod until the salt dissolves completely.
2. Attach the electrical conductivity meter (or set up the bulb circuit) to the solution. Observe whether the bulb lights up or the tester indicates conductivity.
3. Repeat Steps 1 and 2 for the baking powder, cooking oil, slaked lime, and sugar. Use a separate stirring rod for each substance.
4. Record your observations as shown in Table 3.2.
Table 2: Experimental results
Questions
1. Which substance(s) dissolved easily in water? Explain your answer.
2. Which substances are electrovalent? Write their constituent ions.
COVALENT BONDING
Covalent bonding occurs when atoms share electrons to achieve a stable electron configuration, typically similar to that of the nearest noble gas. This type of bond forms when the atoms involved have similar electronegativities (the ability to attract electrons).
Atoms may gain the noble gas electronic structures without becoming ions but by sharing the outermost electrons. For example, a hydrogen atom needs one electron to acquire the helium electronic configuration.
It can combine with another hydrogen atom and share the electrons so that each attain a stable helium configuration. This results in the formation of a hydrogen molecule (Figure 3.4). The kind of the bond formed between the two atoms is called a covalent bond. The covalent bond is a chemical bond formed by the sharing of one or more electrons between atoms.

Covalent bonding in a hydrogen molecule
Note: A single line between two atoms indicates a single covalent bond carrying an electron pair. Two lines are used to represent bonding of two pairs, three lines represent three pairs, and so on. Oxygen requires two electrons to acquire the stable atomic structure. Thus, two atoms of oxygen combine and share four electrons between them (Figure 3.5).

Or, considering only the outermost shells:

Double covalent bond in oxygen molecule
Covalent bonding also occurs between atoms of different elements such as between hydrogen and chlorine. Hydrogen requires one electron to attain a stable helium electronic configuration, while chlorine requires one electron to acquire the argon electronic configuration. The two atoms combine by sharing two electrons between them (Figure 3.6).

Sharing of electrons in a hydrogen chloride molecule
The same happens to the combination between oxygen and hydrogen. Oxygen requires two electrons to acquire a stable structure, while hydrogen requires one electron. Thus, one oxygen atom combines with two hydrogen atoms as shown in Figure 3.7, whereby each hydrogen provides one electron for sharing with oxygen (any pair from the outermost shell).

Covalent bonding in a water molecule
Covalent bonds are usually formed between non-metals. Molecules of covalent compounds are discrete or distinct. This means that the atoms forming the molecules cannot exist freely in the compounds. They remain bound together in molecules and their electrons are not free to form bonds with other atoms.
Properties of covalent compounds
The properties of covalent compounds include the following:
(a) Their melting and boiling points are usually low.
(b) They are usually liquids or gases at room temperatures.
(c) They do not conduct electricity.
(d) They are generally soluble in non-polar solvents such as oil, carbon tetrachloride and hexane.
(e) They are generally insoluble in polar solvents like water.
(f) They can easily vaporise at room temperature.
(g) They have distinct molecular structures.
(h) They are generally non-metallic compounds.
Task 2
Use chemical drawing software such as ChemDraw or ChemSketch to draw the chemical structures for sodium chloride and carbon dioxide based on their molecular formulas. Then, identify the type of bond present in each compound.
Activity 2
Aim: To demonstrate the formation of covalent bonds in molecules
Requirements: Ball-and-stick molecular model kit (or coloured playdough and toothpicks), manila paper, pens, coloured markers and a pair of compasses
Procedure
1. Use different coloured balls or playdough to make spheres representing atoms of hydrogen, oxygen, nitrogen and carbon.
2. Use two hydrogen atoms and one oxygen atom to model a water molecule (H₂O). Connect the hydrogen atoms to the oxygen atom using sticks or toothpicks.
3. For oxygen gas (O₂), connect two oxygen atoms.
4. Use similar steps to create models of methane (CH₄), carbon dioxide (CO₂), and NH₃.
Questions
1. What does a single stick represent?
2. How do atoms in the models share electrons to achieve stable molecules? Indicate the number of shared electron pairs in each molecular model.
VALENCY
Valency refers to the ability of an atom of a given element to combine with other atoms, and is measured by the number of electrons that the atom will donate, receive or share to form a chemical bond. It is the combining power/capacity of an element or a radical.
The combining capacity of an atom of a given element is determined by the number of hydrogen atoms it combines with or displaces. For example, the valency of chlorine is 1 because one atom of hydrogen combines with one atom of chlorine to form hydrogen chloride (HCl). The valency of zinc is 2 because two atoms of hydrogen are displaced from dilute acids by one atom of zinc.
It is easy to predict the valencies of elements from the periodic table. Group I elements have one electron in their outermost shells, and so, their valency is 1. Group II elements have two electrons in their outermost shells, hence their valency is 2.
Valencies are rarely above 4. For elements with more than four electrons in the outermost shells, the valency number is usually obtained by subtracting the number of electrons from eight. For example, sulfur with six electrons in the outermost shell has a valency of 8 – 6 = 2.
Those outermost electrons are called valence electrons. Some elements have more than one valency. For example, iron has valencies of 2 and 3, copper has valencies of 1 and 2, lead has valencies of 2 and 4, and manganese has valencies of 2, 4 and 7. Table 3.3 shows the valencies of some elements and radicals.
Task 3
Use ball and stick models or coloured beads to represent electrons in different shells. Build atoms and determine the valency by observing how many electrons are in the outer shell.
RADICALS
Task 4
Use a chemistry simulation or software to identify, classify and write different formulas of radicals.
A radical is a group of atoms which behaves as a single unit and has a positive or negative charge. It contains at least one unpaired electron. Such a group maintains its identity throughout any chemical reaction.
Most radicals form the non-metallic part of a compound, so their ions are negatively charged. Examples are CO32− and SO42−ions. An exception is for the ammonium radical, NH4+, which behaves like the metallic part of a compound and forms a positive ion.
The valency of the radical is the same as the numerical value that the group acquires when it loses or gains an electron to form an ion. The common radicals with their formulas and valencies are shown in Table BELOW
Table 3: Valencies of some elements and radicals
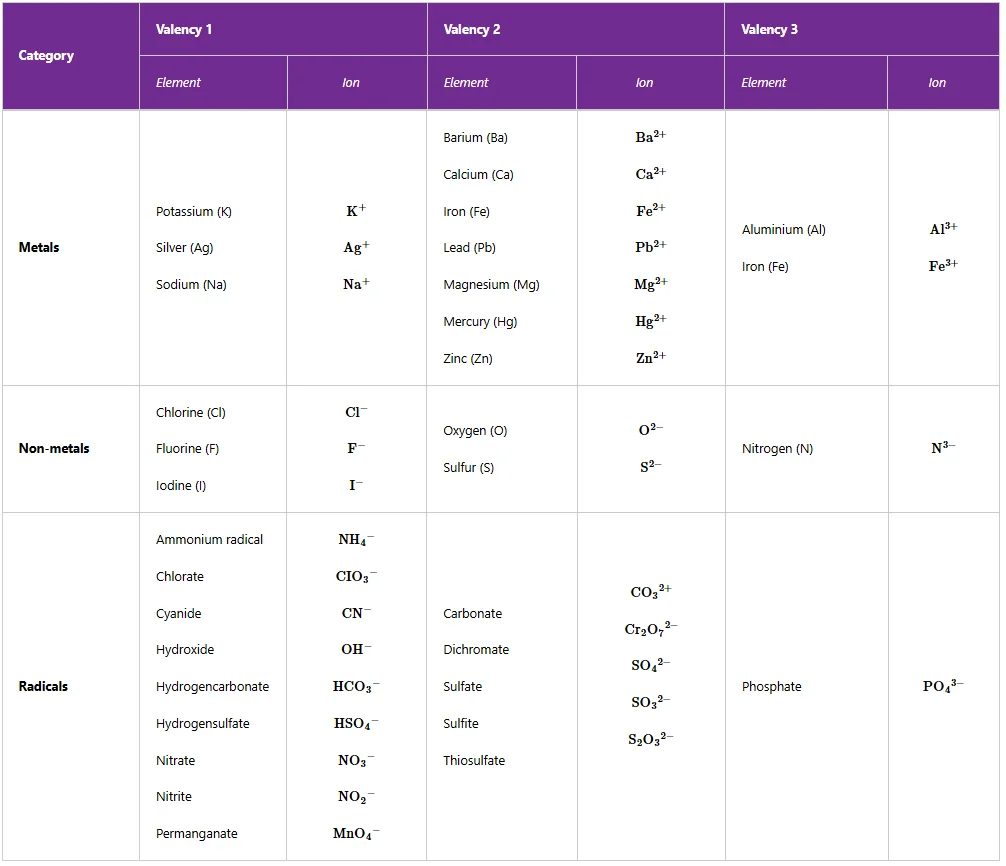
Note: Ammonium radical (NH4+) has a valency of 1 and can react like metals. Its compounds are similar to those of Group I elements.
OXDATION STATE
Oxidation state (also called oxidation number) is the total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom. It is the measure of the electron control that an atom has in a compound compared to the atom in the pure element. The neutral atom has no charge.
Rules for assigning oxidation states
The following are the rules used to assign oxidation states of the elements:
1. The oxidation number of free elements is zero. For example, all elements in the periodic table have the oxidation number of zero.
2. The sum of the oxidation states of all atoms forming a molecule or ion is the net charge of that species. For example, nitrogen (N2), hydrogen (H2) and oxygen (O2) molecules have the oxidation number of zero.
3. In simple ions that consist of only one atom, the oxidation number is equal to the charge on the ion. For example, the oxidation number of a sodium ion (Na+) is +1, aluminium (Al3+) is +3, iron(II) (Fe2+) is +2, and iron(III) (Fe3+) is +3. In an oxide ion (O2−), the oxidation number of oxygen is –2.
4. In their compounds, Group I metals have an oxidation number of +1. Group II metals have an oxidation number of +2, while, Group III metals have an oxidation number of +3.
5. In their compounds, halogens always have an oxidation number of –1.
6. Hydrogen has an oxidation state of +1 in most compounds. The exception is in hydrides of active metals where the oxidation number is –1. For example, the hydrogen atom gains an electron from the lithium atom in lithium hydride (LiH).
7. Oxygen has an oxidation state of –2 when present in most compounds, except:
(a) in peroxides, for example H2O2, where the oxidation number is –1.
(b) when bonded with fluorine to form F2O, the oxidation number is +2.
All oxidation numbers must be consistent with the conservation of charge. This means that for all neutral molecules, the oxidation number of all the atoms must add up to zero. For example, in H2O, two hydrogen atoms each of charge +1 combine with one oxygen atom of charge –2. The charge of the H2O molecule is +2 – 2 = 0.
Note: There is a close relationship between valency and oxidation state. However, they are not the same. Valency is a fixed value, but oxidation state is an arbitrary value (it may vary).
| Example 1 |
Find the oxidation state of chlorine in KClO3
Solution
The oxidation number of potassium is +1
The oxidation number for oxygen is –2
For the three oxygen atoms, the oxidation number is (–2 × 3) = –6
KClO3 is a neutral compound. Therefore, the oxidation number of the compound is zero.
Therefore, + 1 + Cl – 6 = 0
Cl = 6 – 1 = +5
The oxidation number of chlorine in KClO3 is +5.
| Example 2 |
Find the oxidation number of sulfur in SO42−
Solution
The total charge on the sulfate ion is –2
The oxidation number of oxygen is –2
Therefore, S + (–2 × 4) = –2
S – 8 = –2
S = 8 – 2
S = +6
The oxidation state of sulfur in SO42− is +6.
| Example 3 |
Give the oxidation number of Cr in Cr2O72−
Solution
Total charge on the dichromate ion is –2
For oxygen, – 2 × 7 = –14
Therefore, 2Cr – 14 = –2
2Cr = +12
Cr = +6
Therefore, the oxidation number of Cr is +6.
EXERCISE 2
Calculate the oxidation number of each underlined element in the following chemical substances:
1. (a) Na3P―O4 (b) S―O32− (c) N―O2−
2. Explain why ammonia (NH3) is a covalent compound, while ammonium chloride (NH4Cl) is an ionic compound.
CONCEPT OF CHEMICAL FORMULAS
Task 5
Obtain containers such as bottles containing chemicals in the laboratory. Examine them and identify the chemical formulas of the substances on their labels.
A chemical formula is a representation that uses symbols to show the proportions of the elements present in a chemical compound. The number of atoms or groups of atoms are shown by number subscripts.
For example, the chemical formula for sodium sulfide is Na2S, which shows that two atoms of sodium combine with one atom of sulfur to form the molecule of sodium sulfide. For groups of atoms such as radicals, a bracket is used to show that they are being considered as a unit under one valency. For example, in calcium nitrate, Ca(NO3)2, the NO3− radical is in brackets.
There are some points to remember when writing chemical formulas.
1. Positively charged ions (cations) are written before negatively charged ions (anions).
2. A radical must be treated as a unit.
3. Brackets are not used for single elements.
4. The valency 1 is simply assumed and not written in the formula.
The symbols and valencies of the atoms and radicals are important in writing a chemical formula. For example, for arbitrary elements W and X with valencies m and n, respectively, and where X can be a radical or an atom
The following steps can be used to come up with a chemical formula of their compounds:
Step 1: Write the symbols of the elements and radicals, in this case W and X.
Step 2: Write down the ions used, with their valencies as superscripts, that is Wm Xn.
Step 3: Interchange the valencies of W and X and write them as subscripts.

The formula of the chemical compound is WnXm.
Note: When m and n are equal, there is no need for the exchange and, therefore, are not written since they are in a ratio of 1:1.
| Example 4 |
1. Give the formula of the compound of calcium and chlorine atoms.
Step 1: Write down the symbols for calcium or chlorine atoms.
Ca²⁺ Cl–
Step 2: Write down the ions used in the compound with their charges.
Ca²⁺ Cl⁻
Step 3: Write down the ions used with their valencies as superscripts.
Ca2Cl1
Step 4: Interchange the valencies and write them as subscripts.

CaCl₂
The formula of the chemical compound is CaCl₂. The compound contains two chlorine atoms and one calcium atom.
| Example 5 |
Give the formula of the compound of aluminium and sulfate.
Step 1: Al SO₄
Step 2: Al³⁺ SO₄²⁻
Step 3: Al₃SO₄2
Step 4:

Al₂(SO₄)₃
The chemical formula is Al₂ (SO₄)₃.
EXERCISE 3
1. Write the steps used to arrive to the following chemical formulas of the compounds:
(a) MgCl₂(b) Na₂SO₄ (c) NH₄NO₃
2. Write down the chemical formula of each of the compounds formed by the combination of the following elements:
(a) Potassium and chlorine
(b) Calcium and sulfur
(c) Lithium and fluorine
TYPES OF CHEMICAL FORMULAS
Chemical formulas can be divided into three types, namely empirical formula, molecular formula, and structural formula.
An empirical formula is the formula which represents the simplest ratio of the atoms or ions in a compound. The simplest formula is usually determined by considering experimental data. That is why it is called ‘empirical’ which means ‘based on experimentation’. For example, CH₂ shows there are twice as many hydrogen atoms as carbon atoms. It does not show the exact number of each atom of the element in the compound.
A molecular formula shows the actual number of each atom in a molecule. It is a multiple of the empirical formula. For example, if the empirical formula is CH₂, its molecular formula may be C₂H₄, C₄H₈, C₆H₁₂, and so on. Therefore, a molecular formula is equal to n multiplied by the empirical formula, where n is a whole number. Note that when n is 1, the empirical formula equals the molecular formula.
A structural formula is a graphic representation of molecular structure showing how the atoms are arranged. At this level, only the empirical and molecular formulas will be studied.
FORMULA CALCULATIONS
When the percentage compositions of the elements that make up a compound are known, it is possible to obtain both the empirical and molecular formulas of such a compound.
The following are the steps considered when calculating the empirical formula:
Step 1: Obtain the mass of each element in the sample compound. If expressed in percentages, convert the percentage of each of the elements to mass. If the mass or relative molecular mass (R.M.M.) of the compound is not given, an arbitrary mass of 100 g is usually used.
Note: The R.M.M. is the sum of the relative atomic masses (R.A.M.) of all the atoms in a molecule of the compound. The R.A.M. can also be obtained from the periodic table (See Appendix 1).
Step 2: Divide the mass of each element by its R.A.M.
Step 3: Divide each of the values obtained in Step 2 by the lowest value among them.
Step 4: Convert the ratios in Step 3 to whole numbers. These whole numbers give the ratio of each element in the compound.
| Example 6 |
What is the empirical formula for a compound of mass 8.1 g if it consists of 4.9 g of magnesium and 3.2 g of oxygen?
Solution
Step 1: Obtain the mass of each element in the compound. These are already given:
Mass of magnesium = 4.9 g
Mass of oxygen = 3.2 g
Step 2: Divide the mass of each element by its R.A.M.
Magnesium, 4.924 = 0.20
Oxygen, 3.216 = 0.20
Step 3: Divide by the lowest quotient.
Mg : O
0.200.20 : 0.200.20
Step 4: Obtain their whole number ratios directly or by approximation.
Mg : O
1 : 1
The empirical formula is MgO.
| Example 7 |
Given that a certain compound is 69.59% barium, 6.09% carbon and the rest is oxygen. Calculate the empirical formula of this compound.
Solution
Step 1: Assuming that you have 100 g of the compound, then the mass of each element will be:
Ba = 69.59 g
C = 6.09 g
The mass of oxygen will be 100 g – (mass of barium + mass of carbon)
= 100 g – (69.59 g + 6.09 g) = 24.32 g
Note that the 100 g is arbitrary for simplifying the calculations.
Step 2: Divide the mass of each element in the sample by its R.A.M.
Ba = 69.59137.3 = 0.51
C = 6.0912 = 0.51
O = 24.3216 = 1.52
Step 3: Divide each value by the smallest number.
0.510.51 = 1
0.510.51 = 1
1.520.51 = 2.98
Step 4: Obtain the whole number ratios.
Ba : C : O
1 : 1 : 3
The empirical formula is therefore BaCO3.
| Example 8 |
A compound contains 15.8% carbon and 84.2% sulfur. Calculate its empirical formula. If its relative molecular mass is 76, what is its molecular formula?
Solution
Step 1: Assume that you have 100 g of the compound, then find the mass of each element in grams.
C = 15.8 g
S = 84.2 g
Step 2: Divide the mass of each element in the sample by its R.A.M., i.e.
Mass in sampleR.A.M.
C = 15.8 g32 g = 1.32
S = 84.2 g32 g = 2.63
Step 3: Divide throughout by the smallest value, in this case 1.32:
Extra close brace or missing open braceExtra close brace or missing open braceExtra close brace or missing open braceExtra close brace or missing open brace : 2.631.32 = 1:1.99
Step 4: Obtain whole number ratios:
C : S
1 : 2
The empirical formula is therefore CS₂.
The empirical formula together with the relative molecular mass can then be used to establish the molecular formula of the compound.
Molecular formula = n empirical formula, where n is a whole number.
Hint: R.M.M. = n sum of R.A.M.
Therefore, the molecular formula = n empirical formula = n(CS2)
Now, R.M.M. = n sum of R.A.M.
76 = n [12+ (2 × 32)]
76 = n (12 + 64)
76 = 76 n
n = 1
Therefore, the molecular formula is CS₂.
EXERCISE 4
1. A compound has 1.121 g of nitrogen, 0.161 g of hydrogen, 0.480 g of carbon and 0.640 g of oxygen. Deduce its empirical formula.
2. A compound has an empirical formula CH. If it has a relative molecular mass of 78.11, deduce its molecular formula.
3. A compound is analysed and found to contain 52.17% carbon, 13.04% hydrogen, and 34.78% oxygen. Determine its
(a) empirical formula.
(b) molecular formula, if its relative molecular mass is 46.
NOMENCLATURE OF BINARY INORGANIC COMPOUNDS
Everything in the universe bears a name to differentiate it from others. Chemical substances also bear names that range from those of elements to those of compounds. The name of a substance can originate from some factors such as the place of origin, founder, use, and type or classification.
Items or substances that fall under a particular group or classification are named systematically.
A systematic way of assigning names to items that belong to a particular group or classification is called nomenclature.
Scientists use IUPAC nomenclature in naming chemical compounds so that experts and other interested persons around the world understand exactly what the substance is.
BINARY INORGANIC COMPOUNDS
While an inorganic compound is any substance in which two or more chemical elements (usually other than carbon) are combined, always in definite proportions, a binary compound is the one which is formed by two chemical substances. Examples of binary inorganic compounds are CaO, NaCl, and PCl₃.
Inorganic compounds are categorised into ionic and covalent. The nomenclature of ionic compounds differs slightly from that of covalent compounds.
Task 6
Use online resources such as Chemguide to explore detailed explanations and examples of the nomenclature of various binary inorganic compounds. Summarise the key rules for naming these compounds, including binary ionic compounds, covalent compounds and any exceptions.
Nomenclature of binary ionic compounds
Ionic compounds are formed when a metal combines with a non-metal. The following are the steps considered when naming binary ionic compounds:
1. Name the metallic ion that appears first in the formula using the name of the element itself.
2. The second part of the formula which is usually an anion in the compound will end with a suffix “ide”. For example, oxygen becomes oxide, hydrogen becomes hydride and chlorine becomes chloride.
Note:
(a) Some metals always have fixed charges when they form ions, that is,
(i) Group I metals have a charge of +1.
(ii) Group II metals have a charge of +2.
(iii) Group III metals have a charge of +3.
(iv) Silver (Ag) has a charge of +1.
(v) Zinc (Zn) has a charge of +2.
(b) Other metals are multivalent and can thus form more than one ion.
For example; iron (Fe) is bivalent; it has valencies of 2 and 3, copper (Cu) is also bivalent; it has valencies of 1 and 2. Compounds formed from these metals must be distinguished by stating which valency has been used in the compound.
The valency of the respective metal is indicated by capital Roman numbers in parentheses (brackets), for example cobalt(II) chloride, copper(II) oxide and iron(III) oxide. In chemical nomenclature, it is common practice not to leave a space between the name of a metal and its oxidation state written in Roman numbers.
| Example 9 |
What is the name of the compound with the formula FeCl₃?
Solution
The total charge of the molecule is zero and Cl⁻ has a negative charge.
(i) Let x be the charge of Fe
(ii) 1(x) + 3(–1) = 0
(iii) x = +3
(iv) So, the Fe is in the +3 oxidation state. Write the name ‘iron’ and place III in brackets beside it.
(v) Use the name ‘chlorine’ but change the last three letters to “ide”: So the name is iron(III) chloride.
| Example 10 |
What is the name of the compound with the formula CuS?
Solution
(i) Let x be the charge of Cu.
(ii) Sulfur has a charge of –2.
(iii) 1(x)+ 1(–2) = 0
x = +2 for Cu
(iv) Write the name copper and place II in brackets beside it.
(v) Use the name sulfur but change the last two letters to ide. The name of the compound is copper(II) sulfide.
Other examples of the names of binary ionic compounds are:
(a) MgO is named magnesium oxide.
(b) AlCl₃ is named aluminium chloride.
(c) MnO₂ is named manganese(IV) oxide.
Note: Manganese can have more than one charge, but each oxygen ion has a charge of –2. For the compound to be neutral, Mn must have a charge of +4.
Nomenclature of binary covalent compounds
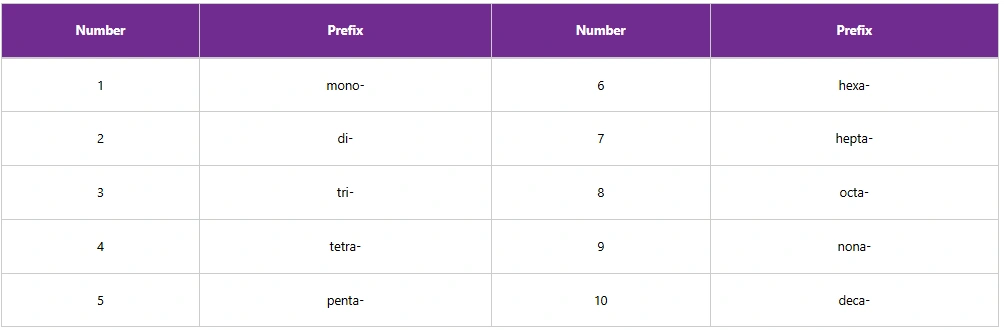
Covalent compounds are formed between two non-metal elements. These compounds are named differently from ionic compounds. The number of atoms are presented by prefixes as shown in Table 3.4.
Table 4: Examples of prefixes
The following are the steps to consider when naming binary covalent compounds:
1. Give the name of the first element.
2. Give the name of the second element with the ending changed to –ide.
3. If more than one compound can form between two elements, use prefixes to indicate the number of atoms of each element.
| Example 11 |
Give the name for PCl₃.
Answer
(i) Since there is one phosphorus atom, use it as the first part of the name.
(ii) There are three chlorine atoms, so use tri in front of chlorine; then, drop the ine in chlorine and replace it with ide.
The name is phosphorus trichloride.
| Example 12 |
What is the name for N₂O₄?
Answer
(i) Use the prefix di in front of nitrogen since there are two atoms.
(ii) Use the prefix tetra in front of the oxygen since there are four atoms.
(iii) Drop –ygen and replace it with ide:
(iv) The name is dinitrogen tetraoxide.
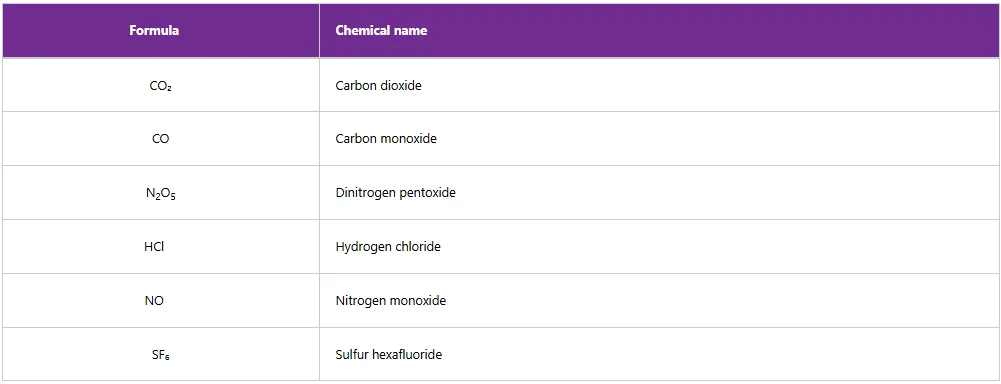
Table 5 gives the formulas and names of some binary covalent compounds.
Table 5: Some binary covalent compounds
Note: IUPAC names of common compounds are given in Appendix 2.
Chemical names of common substances
Chemical names are typically used to provide precise descriptions of substance compositions, including those encountered in daily life. For example, requesting sodium chloride for use in food is uncommon; instead, the term “common salt” is used.
This explains the existence of common names for certain substances. However, it is important to note that some common names are inaccurate and may vary from one place to another. Therefore, they cannot tell the chemical composition of a substance. The chemical names of common substances are provided in Appendix 2.
Activity 3
Aim: To identify and classify the chemical compounds in toothpaste
Requirements: Toothpaste tube (with ingredient list), notebook, pen/pencil, and chart or table for recording observations
Procedure
1. Read the ingredients on the toothpaste tube.
2. Write down the chemical compounds listed in the ingredients.
3. Explore the chemical formulas of the compounds and identify whether each
of the compound contains a metal and a non-metal or only non-metals.
4. Record your findings as indicated in Table 3.6.
Table 6: Chemical compounds present in toothpaste

Questions
(a) In which ways do the ionic and covalent compounds found in toothpaste differ?
(b) Which compound is in the largest amount in the toothpaste?
Chapter summary
1. Chemical bonding involves electrons in the outermost shell of an atom. When the outermost shell is fully filled, the atom is said to be stable.
2. Ions are formed when an atom gains or loses electron(s). Cations are positively charged ions that result from atoms losing one or more electrons. Anions are negatively charged ions that result from atoms gaining one or more electrons.
3. Ionic (electrovalent) bonding usually occurs between a metal and a non-metal. It involves the transfer of electron(s) from the atoms of the metal to the atoms of the non-metal.
4. Covalent bonding takes place between two or more non-metals. It involves atoms of the non-metals sharing electrons that are in their outermost shells.
5. The ability of an atom to combine with other atoms according to the number of electrons it can give, take or share is known as valency.
6. The oxidation state (oxidation number) of an element is the number of electrons that need to be added, shared or removed by its atom, to make a neutral molecule. The oxidation number may be positive, negative or zero.
7. A radical is a group of atoms which behaves as a single unit and has an overall positive or negative charge. A radical can also be an atom, molecule or ion that has unpaired valence electron. Such a group maintains its identity throughout any chemical reaction.
8. A chemical formula is a representation that uses chemical symbols to show the proportions of the elements present in a chemical compound.
9. An empirical formula is the simplest way of writing a chemical formula and indicates the ratio of the atoms in a compound.
10. A molecular formula is a chemical formula that shows the total number of atoms of each element in a molecule of a substance.
11. A systematic way of naming items or substances of a particular category is known as nomenclature.
Revision exercise
Choose the correct answer for Questions 1-11. For other questions, provide the answers as per the demands indicated.
1. What is the valency of Group I elements?
(a) 1 (b) 2 (c) 3 (d) 4
2. What name is given to the force of attraction that holds atoms together to form a molecule?
(a) Chemical change
(b) Chemical bond
(c) Friction
(d) Centripetal force
3. What happens during the formation of ions?
(a) Non-metal atoms gain protons.
(b) Metal atoms lose their outermost electrons.
(c) Metal atoms gain electrons in their outermost shells.
(d) Non-metal atoms lose electrons of their outermost shells.
4. Electrovalent bonding occurs between
(a) metals and metals.
(b) metals and non-metals.
(c) metals and inert gases.
(d) non-metals and non-metals.
5. The following atoms cannot exist freely as single atoms, except
(a) sodium.
(b) chlorine.
(c) argon.
(d) magnesium.
6. A covalent bond is formed due to
(a) opposite charges of atoms.
(b) transfer of electrons of atoms.
(c) forces of attraction between atoms.
(d) sharing of electrons between atoms.
7. Which of the following is a property of covalent compounds?
(a) They do not conduct electricity.
(b) Atoms share only two pairs of electrons.
(c) Mostly are soluble in water.
(d) Their melting and boiling points are very high.
8. What is the oxidation number of nitrogen in NH₄⁺?
(a) +1 (b) –3 (c) +4 (d) +5
9. Which among the following is the correct valency of the phosphateradical?
(a) 5 (b) –3 (c) 3 (d) –5
10. Which of the following sets of symbols represent cations?
(a) K+ and Mg
(b) Mg and Al
(c) Al³⁺ and Cl⁻
(d) K+ and Al³⁺
11. Identify the chemical formula for potassium chlorate(V).
(a) K2ClO3 (b) K(ClO3)2(c) K5ClO3 (d) KClO3
12. Choose the correct formula for the combination of the following ions:
(i) Mg²⁺ and PO₄³⁻
(a) Mg₃(PO₄)₂ (b) Mg₂PO₄ (c) Mg₂(PO)₃ (d) MgPO₄
(ii) Ba2+ and N3-
(a) BaN (b) BaN₂ (c) Ba₂N₃ (d) Ba₃N₂
(iii) Al³⁺ and SO₄²⁻
(a) AlSO₄ (b) Al(SO₄)₂ (c) Al₂(SO₄)₃ (d) Al₃(SO₄)₂
(iv) Zn²⁺ and C₂H₃O₂⁻
(a) ZnC₂H₃O₂ (b) Zn₂C₂H₃O₂ (c) Zn(C₂H₃O₂)₂ (d) Zn₂(C₂H₃O₂)₂
13. (a) Identify the compounds with the incorrect IUPAC names.
(i) KCl – Potassium chloride
(ii) Fe₂(SO₄)₃ – Iron(II) sulfate(VI)
(iii) CaCl₂ – Calcium dichloride
(iv) Na₂SO₄ – Sodium tetraoxosulfate(VI)
(b) Rename the incorrectly named compound(s) in (a) using the IUPAC rules.
14. You are provided with the following list of substances: Mg, MnO₄²⁻, Ca²⁺, Cl, N₂, Cr₂O₇²⁻, Al³⁺, and H₂. Which of these substances are:
(a) atoms?
(b) molecules?
(c) ions?
(d) radicals?
15. A molecule of a certain gas can be represented by the following diagram:

(a) What is the name of the molecule?
(b) What is the molecular formula of the gas?
(c) What type of bonding holds the atoms of the molecule?
(d) Name other five compounds with this type of bonding.
16. Write the electronic configuration of each of the following species:
(a) Aluminum ion
(b) Magnesium
(c) Chloride ion
(d) Neon
17. Name the following compounds:
(a) MgI₂ (b) CCl₄ (c) FeBr₂ (d) CuI₂ (e) H₂S (f) K₂O (g) PCl₅
18. Calculate the oxidation number of Cr in CrO₄²⁻ and Cr₂O₇²⁻ radicals.
19. A compound of sulfur and oxygen is 40.1% sulfur by mass. What is the empirical formula for the compound? The R.A.M. are S = 32.07 and O = 16.00.
20. Write the chemical formulas of the following compounds:
(a) Sodium hydrogen carbonate
(b) Silver trioxonitrate(V)
(c) Copper(I) oxide
(d) Aluminium tetraoxosulfate(VI)
21. Write down any two different ionic states in each of the following elements:
(a) Fe (b) Cu (c) Pb (d) Mn
22. An atom of element X (atomic number 11) and an atom of element Y (atomic number 9) combine to form a compound.
(a) Write the formula of the compound.
(b) State the type of the bond present in the compound. Explain your answer.
23. A hydrocarbon contains 88.88% carbon and 11.12% hydrogen by mass. Calculate the empirical formula of the molecule.
24. A compound consists of calcium 40%, carbon 12% and the rest is oxygen by mass. Determine the empirical formula of the compound.
(a) Calculate the oxidation number of the underlined elements in the following compounds:
(i) N―H4Cl (ii) Na2S―O4 (iii) Al2―O3 (iv) H2O2―
26. Elements T and Q have atomic numbers 12 and 17, respectively. Use the two elements to answer the following questions:
(a) Write the electronic configuration of element Q.
(b) What is the valency of element T?
(c) Write the chemical formula of a compound formed when T and Q combine.
(d) Giving a reason predict the type of the bond formed by the combination of elements T and Q.
(e) In which group and period in the periodic table do elements Q and T belong?
27. An organic compound contains 26.70% carbon, 2.20% hydrogen and 71.10% oxygen. If its mass is 90 g, determine its molecular formula.