TOPIC 2 PERIODIC CLASSIFICATION – CHEMISTRY FORM TWO
Introduction
Many elements have been discovered. Therefore, studying each element individually is challenging. The elements are better understood and managed when they are classified. The most effective way to classify them is by using a table known as the periodic table.
In this chapter, you will learn about the development of the periodic table, electronic configuration and element positioning in the periodic table, and changes in physical and chemical properties across periods and down the groups. The competencies developed will provide a foundation for studying elements and predicting how chemicals behave and their applications in everyday life.
Think: Periodic classification is the backbone of chemistry applications.
I. Development of the periodic table
Task 1
Utilise online or any reliable resources to search the dynamic changes that led to the development of the modern periodic table.
For a long time, chemists have used various ways of grouping elements with similar properties. The simplest of these has been classifying elements as either metals or non-metals. In 1866, a British chemist, John Newlands, thought of the idea of arranging elements in order of their increasing atomic masses. Newlands arranged the elements according to Table below
Table 1: Newlands’ first arrangement of elements
|
H |
Li |
Be |
B |
C |
N |
O |
F |
Na |
Mg |
Al |
Si |
P |
S |
Cl |
K |
Ca |
Newlands noticed that an element tends to display characteristics similar to the 8th element in front of it. He arranged the elements in columns according to a law he called the Law of Octaves (Table 2.2). However, his classification was unfortunate since he grouped together certain elements which had different characteristics. For example, oxygen (O) was placed in the same group as iron (Fe) and sulfur (S). Newlands’ ideas were therefore rejected by many scientists.
Table 2: Newlands’ octaves of elements
|
H |
Li |
Be |
B |
C |
N |
O |
|
F |
Na |
Mg |
Al |
Si |
P |
S |
|
Cl |
K |
Ca |
Cr |
Ti |
Mn |
Fe |
A Russian chemist, Dimitri Mendeleev, later improved Newlands’ ideas and convinced other chemists to use them. He intended to illustrate recurring trends (periodic trends) in the properties of elements.
In 1869, Mendeleev summarised his Periodic Law which states that the properties of elements are a periodic function of their relative atomic masses. He arranged elements according to their increasing atomic masses and by the similarity of properties.
This resulted in an early version of the periodic table of elements (Table BELOW). A vertical column of elements is called a group and a horizontal row is called a period. He, however, left gaps in the table predicting that there were existing elements yet to be discovered.
Table 3: Part of Mendeleev’s periodic table

II. Modern periodic table
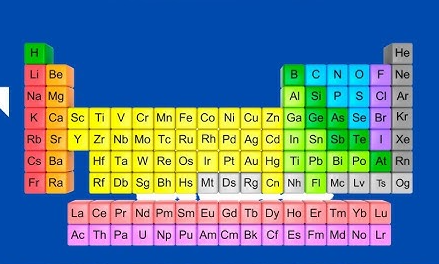
The modern periodic table is a systematic arrangement of elements in order of increasing atomic numbers. It is a result of several modifications to Mendeleev’s periodic table as new elements were discovered and scientific theories advanced to explain the chemical behaviour of elements.
Unlike Mendeleev’s Periodic Law, which was based on atomic masses, the modern periodic table is structured according to atomic numbers. Therefore, it led to the development of the modern Periodic Law which states that the properties of elements are a periodic function of their atomic numbers.
This means that elements exhibit recurring (periodic) chemical properties when arranged in increasing atomic number, a phenomenon known as periodicity. For example, lithium, a very reactive metal, has one electron in its outermost shell.
The eighth element after lithium is sodium, which shares many of the same characteristics as lithium, including a single electron in its outermost shell and high reactivity. Similarly, the eighth element after sodium is potassium, which also exhibits the same properties. This periodic recurrence of chemical behaviour is among the fundamental principles of the modern periodic table.

Note: In the periodic table, groups are shown using Roman numerals from I to VIII/0. These numbers indicate how many electrons are in the outermost shell of an element. Using Roman numerals helps make it easier to understand how elements react and form bonds.
However, the modern International Union of Pure and Applied Chemistry (IUPAC) system, adopted worldwide, numbers groups from 1 to 18 consecutively. This standardised format aims to facilitate group identification and promote consistency in educational and scientific contexts. Table 2.4 shows the first twenty elements in the periodic table.
Table 4: Position and electronic arrangements of the first twenty elements in the periodic table

Exercise 1
1. Why were the noble gases not included in Mendeleev’s periodic table?
2. How does the electronic configuration of nitrogen compare to that of phosphorus?
3. Explain the significance of the Law of Octaves in understanding periodic trends.
4. Mendeleev left gaps in his periodic table. Predict how these gaps demonstrate his understanding of properties of elements.
5. Mendeleev used atomic masses to arrange elements. If you were to use atomic numbers, how would this impact the organisation of the periodic table?
6. How did the Mendeleev’s and Newlands’ works contribute to understanding the relationships between elements’ physical and chemical properties.
III. Electronic configuration and element positioning in the periodic table
Task 2
Use reliable online resources to analyse the relationships between electronic configurations and the positions of elements in the periodic table.
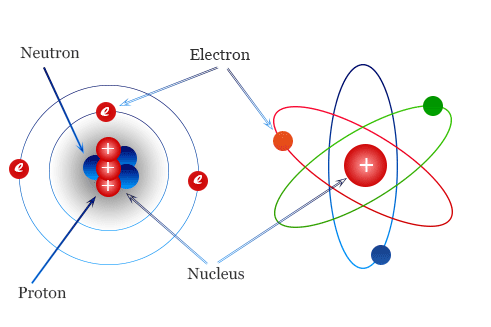
The arrangement of electrons in an atom follows a specific pattern known as electronic configuration. This configuration determines the positioning of elements in the periodic table and their properties. Elements with similar valence electron configurations share common properties and belong to the same group in the periodic table.
Elements are arranged in groups based on the number of electrons in their outermost shells. Elements in the same group exhibit similar chemical properties because they have the same number of valence electrons. Valence electrons are the outermost electrons of an atom that are involved in forming chemical bonds. The groups are labelled using Roman numerals (I to VIII), with the group number corresponding to the number of electrons in the outermost shell as shown in Figure 2.1.
The elements in Group I (except hydrogen) are called, alkali metals because they react with water to form an alkaline solution. These elements are lithium (Li), sodium (Na), and potassium (K), each having one electron in the outermost shell. Note that, even though hydrogen is placed in Group I, it is not an alkali metal. It carries some properties which are similar to those of Group I elements and others which are similar to those of Group VII elements. Therefore, some periodic tables such as Newlands’ place it in the same group as Group VII elements, such as fluorine and chlorine.
Group II elements are called alkaline earth metals. They have properties similar to those of Group I elements, but they are less reactive. Group II elements also form alkaline solutions when they react with water. Their oxides are stable, insoluble solids that historically were referred to as “earths,” giving the term earth
Group VII elements are called halogens (’salt formers’ in Greek) because they react with metals to form compounds called salts. Group VIII elements are usually referred to as Group 0 elements. They are called noble gases and all their shells are completely filled with electrons. They were formerly called inert gases because they do not readily react to form compounds.
The elements in the block between Group II and Group III are transition elements. These elements have high densities and melting points, form coloured compounds, and often act as catalysts. Metalloid elements such as boron (B), silicon (Si), and germanium (Ge) exhibit both metallic and non-metallic properties.

Sketch of the periodic table showing groups of elements
Elements are also arranged into periods, which indicate the number of electron shells in an atom. Table 2.4 shows that elements with the same number of shells belong to the same period. Periods are numbered from 1 to 7. The electronic configuration entails the number of shells for each element. Periods and number of shells of the first twenty elements are shown in Table 2.5.
Table 5: Periods and number of shells of the first twenty elements
| Period | Elements | Number of shells |
| Period 1 | Hydrogen and helium | 1 |
| Period 2 | Lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine and neon | 2 |
| Period 3 | Sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine and argon | 3 |
| Period 4 | Potassium and calcium | 4 |
Electronic configurations help to explain the positioning of elements in the periodic table. Elements in the same group share the same number of valence electrons, while elements in the same period have the same number of electron shells. This arrangement forms the basis for predicting chemical behaviour and classifying elements efficiently.
Exercise 2
1. Write the electronic configuration and indicate the group and period for each element in the table.
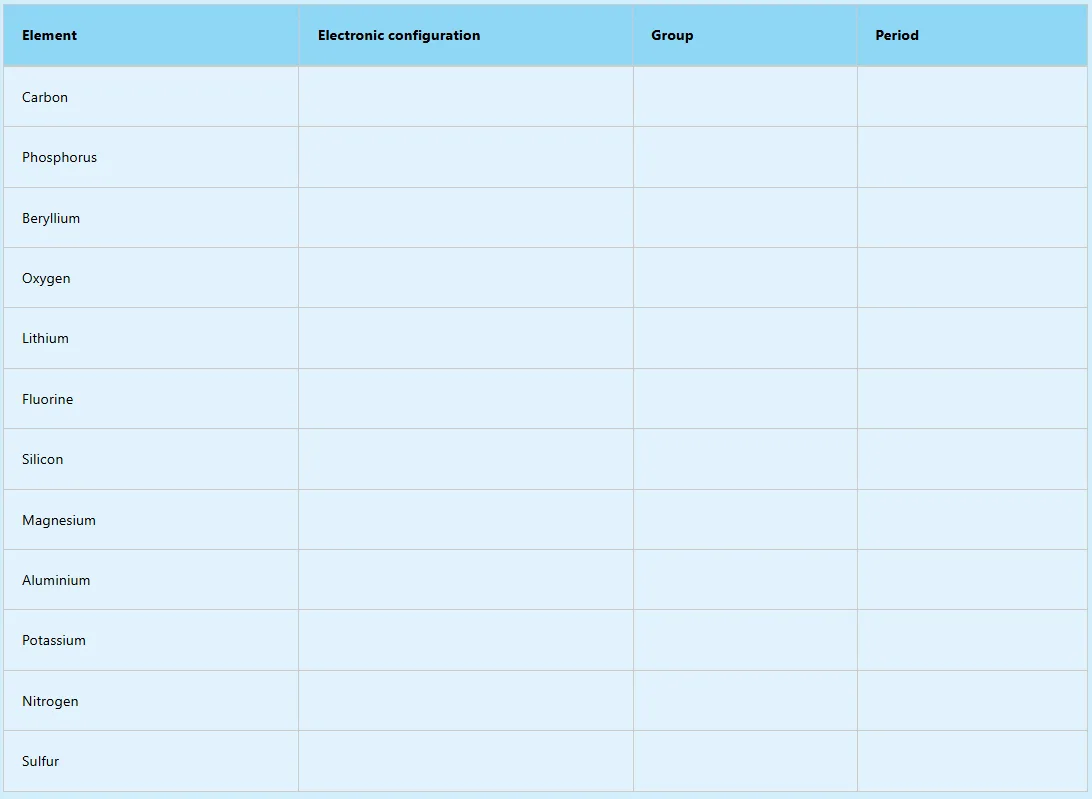
2. Argon and sodium are in the same period but behave differently. Use their electronic configurations to justify their differences.
3. How does an element’s electronic configuration determine its position in the periodic table? Provide examples.
4. Why do elements in the same group of the periodic table have similar chemical properties?
5. Predict the position of an element with the electronic configuration 2: 8: 3 in the periodic table and justify your answer.
Task 3
Sketch a modern periodic table on manila paper using a pencil and ruler, ensuring clear rows for periods and columns for groups. Use different coloured marker pens to assign a distinct colour to each group. Then, fill each block with the chemical symbol, atomic number, and relative atomic mass of the corresponding elements. Select one element from each group and write its electronic configuration below its corresponding block to illustrate its relationship with its position in the periodic table.
IV. Trends in physical and chemical properties across periods and down groups
Task 4
Utilise reliable online resources to search and analyse trends in the periodic table and describe how the properties of elements vary across the periods and down the groups.
The periodic table is a systematic arrangement of elements that helps in understanding patterns in their physical and chemical properties. Elements in the same row (period) and the same column (group) exhibit trends in melting points, boiling points, density, electronegativity, ionisation energy, atomic size, and reactivity. Recognising these trends is essential for applications in various settings, including home settings and industries such as material manufacturing, energy storage, and healthcare.
Melting point is the temperature at which a solid turns into a liquid. For example, ice melts into water when exposed to heat, such as on a hot day. On the other hand, boiling point is the temperature at which a liquid changes into a gas, for instance water boiling and turning into steam when heated on a stove. Density, another important property, refers to the mass per unit volume of a substance and reflects its compactness.
Electronegativity is the tendency of an atom to attract shared electrons in a chemical bond toward itself. This property plays a crucial role in interactions, such as those between the ions of salt and water molecules when the salt dissolves in water. On the other hand, ionisation energy is the energy required to remove an electron from an atom or ion in its gaseous state. Another fundamental property is the atomic size or radius, which measures the distance from an atom’s nucleus to its outermost electron shell.
Reactivity refers to the ability of an atom of a given element to interact chemically with other substances. For instance, alkali metals (Group 1) are highly reactive due to their tendency to lose one electron, whereas noble gases (Group VIII) exhibit minimal reactivity because of their stable electronic configurations. Understanding these properties and their trends helps to explain the behaviour of elements in reactions and their industrial applications.
V. Trends in physical properties across periods
Task 5
Use an interactive simulation or any reliable resources to explore the trends in physical properties across periods of the periodic table. Analyse these trends and explain their practical applications in real-life scenarios.
The following are trends in physical properties across periods:
(a) Decrease in atomic radius
Atomic radii decrease from left to right across a period due to the increasing nuclear charge, which pulls the electrons closer to the nucleus. This trend contributes to designing strong and durable materials in construction and manufacturing, as smaller atomic sizes promote stronger atomic bonding and improved structural strength.
(b) Transition from metallic to non-metallic properties
Metals dominate the left side of the periodic table, while non-metals are positioned on the right. On moving across a period, metallic properties decrease, and non-metallic properties increase. This trend influences material selection, with metals such as iron and aluminium serving as structural materials due to their strengths, while non-metals such as carbon, silicon, sulfur, and chlorine in plastics and rubber function as insulating materials because of their poor conductivity.
(c) Variations in physical states
Elements exist in different states at room temperature (25 °C). Metals such as aluminium and iron remain solid, elements such as oxygen and nitrogen exist in gaseous form, and a few, including bromine, are liquids. This trend is essential for safe storage and transportation. For example, gases require pressurised cylinders, while solids remain stable in containers.
(d) Variations in melting and boiling points
Across a period, melting and boiling points generally increase at first due to stronger metallic or covalent bonding. However, they begin to decrease after Group IV as elements start to form molecules held together by weaker intermolecular forces. Typically, the melting and boiling points increase from Group I to Group IV, then decrease from Group V and stay relatively low to Group VIII.
This trend has several practical applications in everyday life and industry. For example, metals such as sodium (Na), magnesium (Mg), and aluminium (Al), which lie between Groups I and III, have relatively high melting points due to strong metallic bonding. These properties make them suitable for use in cooking utensils, building materials, and aircraft parts where heat resistance is important.
Elements like silicon (Si) in Group IV, with a giant covalent structure and very high melting point, are essential in the manufacture of computer chips and solar panels. On the other hand, elements found in Groups V to VIII, such as nitrogen (N₂), oxygen (O₂), and neon (Ne), exist as simple molecules with low melting and boiling points. These are commonly used in gas form; for example, oxygen is used in hospitals for respiration, nitrogen in food preservation, and neon in lighting systems.
(e) Increase in ionisation energy
Ionisation energy increases from left to right across a period due to stronger nuclear attraction, making it harder to remove electrons from an atom. Elements with high ionisation energy, such as noble gases, are used in lighting and insulation due to their chemical stability.
VI. Trends in chemical properties across periods
The following are changes in chemical properties across periods:
(a) Increase in electronegativity
Electronegativity increases from left to right across a period, with non-metals attracting electrons more strongly. This is due to the stronger nuclear attraction resulting from the increasing number of protons in the nucleus. This trend is particularly crucial in various applications, such as semiconductor technology, where elements like silicon and germanium are widely used in electronic devices.
(b) Decrease in metallic character
Metallic characters decrease, and non-metallic characters increase across a period. Metals tend to lose electrons, while non-metals gain electrons. This trend is important in various applications such as battery production, where metals act as electron donors.
(c) Variations in chemical reactivity
Metals on the left are highly reactive and lose electrons easily, while non-metals on the right become more reactive in gaining electrons. For metals, reactivity decreases from left to right, while for non-metals, reactivity increases from left to right. This trend plays crucial roles in different activities such as drug formulation and material design, ensuring the stability and effectiveness of compounds in pharmaceuticals and engineering.
VII. Trends in physical properties down a group
(a) Atomic size increases down a group as more electron shells are added.
This expansion causes atoms to become larger, affecting their physical behaviour. Larger atomic size influences material performance under high-pressure environments, making certain elements suitable for deep-sea applications and industrial machineries.
(b) Density also increases down a group as atomic mass increases more significantly than the atomic volume.
Heavier elements tend to have stronger structural properties, making them valuable in industries that require durability and strength. These elements play crucial roles in construction and aerospace engineering, where materials must withstand extreme conditions.
(c) Melting points for metals generally decrease down a group due to weaker metallic bonding.
As atomic sizes increase, the attraction between metal atoms weakens, reducing the energy required to melt the substances. This trend is significant in the design of alloys for safety devices such as fuses, which need to melt easily to prevent electrical hazards.
(d) Ionisation energy decreases down a group in the periodic table.
This is because as atomic sizes increase, the outermost electrons are farther from the nucleus. As a result, the attractions between the nucleus and outer electrons become weaker, making it easier for metals to lose electrons. The trend in ionisation energy influences an element’s reactivity, the types of compounds it forms, its electrical conductivity, and its role in biological and industrial processes such as metallurgy, battery design, and semiconductor manufacturing.
VIII. Trends in chemical properties down a group
(a) Electronegativity decreases down a group.
This is because, as atomic size increases, the outer electrons are farther from the nucleus. This condition reduces the attractions between the nucleus and electrons, making atoms less able to attract electrons in a chemical bond.
(b) Metallic character increases down a group.
These properties are crucial in selecting metals for use in catalysts and chemical processing industries. Reactivity trends differ between metals and non-metals as you move down a group. Metals become more reactive because they are more likely to lose electrons, which is advantageous in processes such as metal extraction from ores. In contrast, non-metals become less reactive because their ability to attract electrons weakens. This behaviour is applied in industries where reactive metals facilitate the breakdown of substances as in cleaning agents, while non-metals help stabilise compounds to prevent undesired reactions.
IX. Trends in selected groups
Groups I, II, and VII elements exhibit distinct physical and chemical properties due to their unique positions in the periodic table. Group I (alkali metals) and Group II (alkaline earth metals) are highly reactive metals, while Group VII (halogens) consists of reactive non-metals.
Their physical properties, such as atomic radius, ionisation energy, density, and melting points, exhibit clear trends within each group, as shown in Tables 2.6 and 2.7. Chemically, they differ in reactivity and electronegativity, reflecting variations in their electronic configurations. Understanding these properties provides valuable insights into their behaviour and applications in chemistry and other fields.
Group I: Alkali metals
Group I consists of metals such as lithium (Li), sodium (Na), potassium (K), rubidium (Rb), and caesium (Cs) as shown in the Table 2.6. The data from this table show that the physical and chemical properties of these elements generally increase or decrease down the group. Each of these elements has one electron in its outermost shell. Lithium, sodium and potassium react very readily with water or air and are stored in oil.
Table 6: Trends in Group I
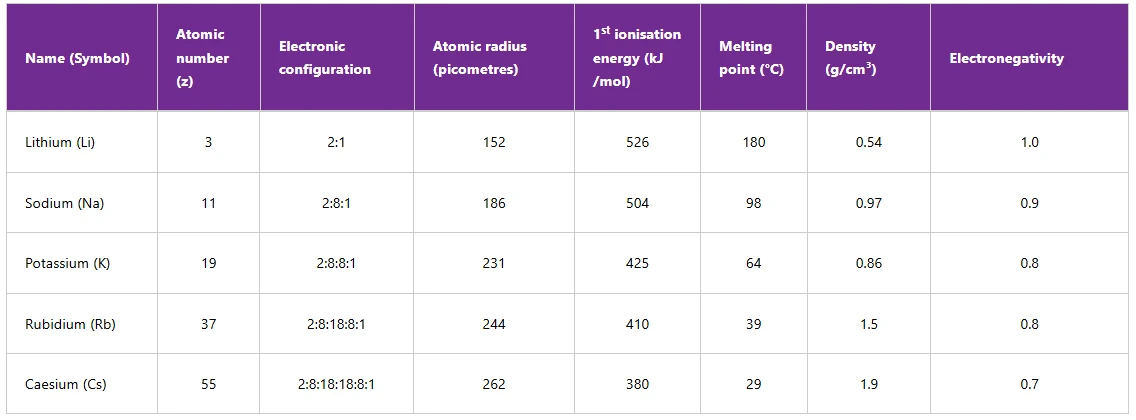
Note: 1. Francium (Fr) is also an alkali metal but is rarely discussed or experimented in the discussion or experiments involving Group I elements due to its radioactive nature. It is also among the rarest naturally occurring elements.
2. The way electronic configurations of rubidium and caesium are written, is above the scope of this book. However, for the sake of understanding this section, the electronic configurations are written to show the period number, group number, and their relations to the trends.
The Group I elements have the following properties:
Physical properties
(a) They are good conductors of heat and electricity.
(b) They are soft metals.
(c) They have low density.
(d) They have shiny surfaces when freshly cut.
Chemical properties
(a) They burn in oxygen or air with a characteristic flame colour to form white solid oxides. These oxides dissolve in water to form alkaline solutions of the metal hydroxides.
Metal + Oxygen —–> Metal Oxide
(b) They react vigorously with water to give alkaline solutions and hydrogen gas.
Metal + Water ——> Metal Hydroxide + Hydrogen gas
Task 6
Utilise educational resources to explore the practical applications of Group I elements such as lithium in batteries or sodium in table salt. Prepare a poster or presentation linking their physical and chemical properties (for example, reactivity and conductivity) to everyday uses.
Activity 1
Aim: To investigate the reactivity of Group I elements (alkali metals) down the group
Requirements: Small pieces (equivalent to the head of a matchstick) of lithium (Li), sodium (Na), and potassium (K), three beakers, water, phenolphthalein or universal indicator, forceps or tongs, watch glass, dropper, safety goggles and gloves
Caution: Handle Group I elements cautiously by using small quantities, wearing protective gear, working in a controlled environment, avoiding direct contact, and keeping a fire extinguisher nearby to prevent accidents during their vigorous reactions with water.
Procedure
1. Fill three beakers halfway with water.
2. Add a few drops of phenolphthalein or universal indicator to each beaker.
3. Using forceps, carefully drop a small piece of lithium into the first beaker and observe the reaction.
4. Repeat Step 3 for sodium and potassium in separate beakers.
5. Record observations for each metal, focusing on the rate of reaction, colour change (due to the indicator), production of gas and sound.
6. Clean up all the apparatus safely under the supervision of the teacher.
Questions
1. Which element was the fastest to react with water?
2. How did the colour of water change and what did it indicate?
3. What trend did you observe in the reactivity of those elements down the group?
4. How does this experiment support the idea of periodic classification?
Chapter summary
1. The periodic table of elements is a method of displaying chemical elements in a table format. It was developed after several modifications to the Mendeleev’s periodic table.
2. Mendeleev’s Periodic Law states that the properties of elements are periodic functions of their relative atomic masses.
3. The Modern Periodic Law states that the properties of elements change systematically according to their atomic numbers.
4. Periodicity refers to the regular periodic changes of properties of elements due to changes in atomic numbers.
5. Elements with the same number of electrons in their outermost shells belong to the same group.
6. The group number signifies the number of electrons in the outermost shell of an element.
7. Elements with the same number of shells belong to the same period.
8. The electronic configuration of an element entails the electronic arrangements in the shell(s), number of shells, and the group to which the element belongs.
Revision exercise
Choose the correct answer for Questions 1-7. For other questions, provide the answers as per the demands indicated.
1. Non-metals are generally better ________ than metals.
(a) conductors of electricity
(b) brittle materials
(c) malleable materials
(d) conductors of heat
2. The electronic arrangement of an element is 2: 3. This element is in _______ of the periodic table.
(a) Group II
(b) Group VIII
(c) Period 3
(d) Period 2
3. The following statements describe the alkaline earth metals except they
(a) burn in oxygen to form solid white oxides.
(b) become less reactive down the group.
(c) are good conductors of heat and electricity.
(d) react with water.
4. An element with electronic configuration of 2: 8: 3 belongs to Group ___ and period ____ of the periodic table.
(a) III … 2
(b) III … 3
(c) II … 4
(d) II … 5
5. Identify the electronic configuration of an element with 16 electrons.
(a) 4: 8: 4
(b) 3: 8: 5
(c) 2: 8: 6
(d) 2: 10: 4
6. Hydrogen is placed in Group I elements because it
(a) is in period 1.
(b) loses an electron.
(c) is an inert gas.
(d) has atomic mass of 1.
7. Group I elements burn in oxygen to form
(a) metal oxides.
(b) non-metal oxides.
(c) hydroxides.
(d) carbon dioxide.
8. (a) A particular metal reacts slowly with water to give a strong alkaline solution. In which group of the periodic table should it be placed? Give a reason.
(b) What is common about the noble gases regarding the following properties?
(i) Electronic arrangements
(ii) Chemical reactions
9. Why are certain elements in the periodic table referred to as metalloids?
10. The following diagram represents the parts of the periodic table with four areas denoted by 1, 2, 3 and 4:

(a) Which part contains mostly non-metals?
(b) Which part contains most elements whose oxides dissolve in water?
(c) Which part contains transition elements?
(d) Which part mostly contains elements with both metallic and non-metallic characteristics?
11. (a) Given the elements calcium, sulfur, chlorine, helium and neon, write their:
(i) period numbers.
(ii) group numbers.
(iii) atomic numbers.
(iv) number of electrons in their atoms.
(v) electronic configurations.
(b) Which of the above elements would you expect to have similar properties? Give reasons.
12. During a science lesson, students added a small piece of sodium (Na) into water and observed fizzing and heat. They repeated the same with magnesium (Mg), and there was very little reaction. Using periodic classification, explain why the reaction of sodium with water was vigorous than that of magnesium.
13. Greenish Secondary School is building a simple application (App) that can help Form Two students to identify if an element is a metal, non-metal, or metalloid. What features of periodic classification should be included in the App to help students make the correct decision?
14. A cook wants to buy a new cooking pot. The shopkeeper offers two options: one made from aluminium and the other from a shiny, brittle material. Based on periodic classification, which material is better for cooking pots and why?
15. Form Two students are curious about the elements used in smartphones. They found that lithium (Li) is used in the battery and silicon (Si) in the screen. Based on their positions in the periodic table, explain why these elements are suitable for their specific roles in smartphones.
16. Magnesium has the electronic configuration 2: 8: 2. Explain how this configuration relates to its chemical reactivity and its position in the periodic table.
17. Two elements, A and B, have electronic configurations 2: 8: 8 and 2: 8: 2, respectively. Compare their positions in the periodic table and predict which is more reactive. Give reasons for your answers.
18. Predict the position of an element in the periodic table with the electronic configuration 2: 8: 8: 1. Justify your answer. What chemical properties would you expect this element to have?
19. Element R belongs to period 3 and Group VI in the periodic table.
(a) Draw its atomic structure.
(b) Determine its atomic number.
20. The following diagrams show the structures of atoms of elements X and Y:

(a) Which of the elements is stable?
(b) Which of the two elements is chemically more reactive? Explain.
(c) Identify elements X and Y. Give the criteria you used.
21. How does the concept of periodicity in the periodic table aid modern chemists in organising information about chemical elements?
22. What happens when alkali earth metals burn in oxygen?
23. Consider elements 11T and 17Q. Which of the two elements is more electronegative? Explain.