TOPIC 5 ACIDS, BASES, AND SALTS – CHEMISTRY FORM TWO
Introduction
Acids, bases, and salts are essential for the proper functioning of our bodies and the health of the environment. Medicines, foodstuffs, and fertilisers are among the substances that contain acids, bases, or salts.
In this chapter, you will learn about acids, bases, acid-base indicators, salts, their properties, and uses. The competencies developed will enable you to prepare and use acids, bases, acid-base indicators and salts, as well as apply the concept of acid-base neutralisation in daily life.
Think: The usefulness of acids, bases and salts in daily life
ACIDS
Task 1
Use reliable online simulations, and other resources to explore the concepts of acids and their applications in daily life.
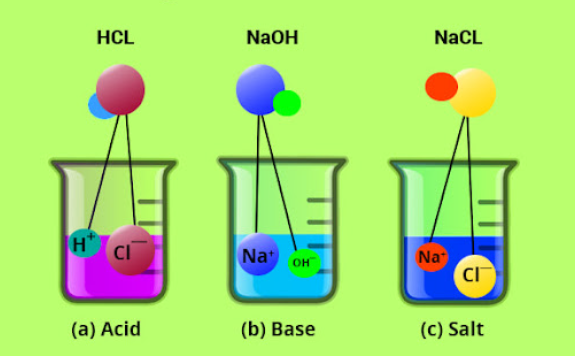
An acid is any substance which produces hydrogen ions (H+) in water as the positively charged ions. In this case, acids are substances made of hydrogen ions and other ions.
Some acids occur naturally, but others are synthesised (do not occur naturally). Therefore, acids can be categorised as natural acids and synthetic acids. The substances which contain natural acids include sour milk and citrus fruits, such as oranges, lemons, and limes. Figure 5.1 and Table 5.1 show some natural sources of acids.




Some fruits with natural sources of acids
Table 1: Examples of natural acids and their sources

On the other hand, synthetic acids are artificially produced acids. Examples of synthetic acids include sulfuric acid, nitric acid and hydrochloric acid.
Acids can be classified as mineral (inorganic) acids and organic acids.
Mineral acids are synthesised in industries from inorganic substances, while organic acids are synthesised in industries or obtained directly from natural organic materials.
Examples of mineral acids include nitric acid (HNO₃), sulfuric acid (H₂SO₄), hydrochloric acid (HCl), carbonic acid (H₂CO₃), phosphoric acid (H₃PO₄), nitrous acid (HNO₂), and sulfurous acid (H₂SO₃).
Examples of organic acids include ethanoic acid/acetic acid (CH₃COOH) and methanoic acid (HCOOH). The most common acids that are found in a chemistry laboratory include hydrochloric acid, sulfuric acid, nitric acid, and acetic acid.
.Common acids in the laboratory are stored in various containers such as plastic bottles or glass bottles as shown in Figure below

PROPERTIES OF ACIDS
Acids have certain physical and chemical properties.
A. Physical properties of acids
The following are the physical properties of acids:
1. Acids have a sour taste: A sour taste of vinegar, sour milk, and many unripe fruits such as lemons, is caused by the acids present in them.
2. Most acids are water soluble: They can be diluted with water to reduce the intensity of their acidity. Diluted acids feel watery unlike concentrated ones.
3. Acids have the ability to conduct electricity: When acids are dissolved in water, they release ions which conduct electricity. The strength of acids as electrolytes varies; strong acids ionise completely, while weak acids ionise partially. This is why some acid solutions are commonly used in car and solar batteries.
4. Acids have varied boiling and melting points, depending on their molecular structures. Some acids have high boiling points, for example, sulfuric acid (337 °C), whereas others have low boiling points, for example formic acid (100.8 °C).
5. Acids are corrosive in nature: Acids can have a corrosive action on various substances such as paper as well as clothes. They can also damage or even destroy metals. Therefore, they should be handled with extreme care; otherwise they can corrode the skin.
6. Acids are mostly present in liquid or gaseous forms, but there are some which exist in solid form such as oxalic acid.
7. Acids change the colour of phenolphthalein indicator to colourless.
8. Acids change the colour of methyl orange indicator to red.
B. Chemical properties of acids
Acids have various chemical properties, including the following:
1. Acids have a pH less than 7 on a pH scale. Low pH values indicate high acidity, for example, hydrochloric acid (HCl) has a pH between 1.5 and 3.5, nitric acid (HNO₃) has pH around 3, and sulfuric acid (H₂SO₄) has pH between 2 and 3.
2. Solution of an acid turns blue litmus paper red (Figure below). This is used in the identification of acids.

Blue litmus paper immersed in an acid solution
3. Acids react with carbonates and hydrogencarbonates to produce salt, water and carbon dioxide gas as shown in the following chemical equations:
2HCl(aq)+CaCO3(s)⟶CaCl2(aq)+H2O(l)+CO2(g)
H2SO4(aq)+Ca(HCO3)2(aq)⟶CaSO4(s)+2H2O(l)+2CO2(g)
One test of the produced gas (carbon dioxide) is to bubble it through limewater. The limewater will turn milky or cloudy white. The laboratory experimental set-up for the reactions of acids and carbonates is shown in Figure below.

Set-up for the reaction of acids with carbonates
4. Acids react with metals above hydrogen in the reactivity series to form salt and hydrogen gas. Some examples are shown in the following equations:
Mg(s)+H2SO4(aq)⟶MgSO4(aq)+H2(g)
Zn(s)+2HCl(aq)⟶ZnCl2(aq)+H2(g)
Mg(s)+2HNO3(aq)⟶Mg(NO3)2(aq)+H2(g)
Note:
(a) The reaction between lead and dilute hydrochloric acid or sulfuric acid forms an insoluble coating of lead chloride and lead sulfate, respectively which prevents further reaction. The two reactions are very slow and the process may take hours before a few bubbles of hydrogen gas are observed. Copper does not react with dilute hydrochloric acid and sulfuric acid because it is below hydrogen in the reactivity series of metals.
(b) The reactions of dilute nitric acid and most metals produce water instead of hydrogen gas. This is because nitric acid is a strong oxidising agent. Its oxidising power depends on the concentration of the acid; the higher the concentration of acid, the higher the oxidising power of the acid. The following equations present the reaction between metals and concentrated nitric acid:
Pb(s)+4HNO3(l)⟶Pb(NO3)2(aq)+2H2O(l)+2NO2(g)
Ca(s)+4HNO3(l)⟶Ca(NO3)2(aq)+2H2O(l)+2NO2(g)
5. Acids react with metal oxides or hydroxides to form salt and water only, as shown in the following examples:
CuO(s)+2HNO3(aq)⟶Cu(NO3)2(aq)+H2O(l)
Ca(OH)2(aq)+2HNO3(aq)⟶Ca(NO3)2(aq)+2H2O(l)
Activity 1
Aim: To investigate the reactions of dilute acids with metals
Requirements: Test tubes, test tube rack, retort stand and clamp, wooden splints, lighter, 2 M sulfuric acid, 2 M hydrochloric acid, 2 M nitric acid, zinc, magnesium ribbon, lead, and copper
Procedure
1. Transfer about 5 cm³ of 2 M sulfuric acid, 2 M nitric acid, and 2 M hydrochloric acid in three separate test tubes.
2. Add a small piece of zinc metal to each test tube, one at a time, and record your observations.
3. Put a lighted splint at the mouth of each test tube, one at a time, as shown in Figure 5.5, and record the observations.
4. Repeat Steps 2–3 with magnesium, lead, and copper, each at a time instead of zinc metal.

Set-up for the reaction of dilute acids with metals
Questions
1. What are the balanced chemical equations for the reaction between:
(a) zinc and dilute hydrochloric acid?
(b) zinc and dilute sulfuric acid?
(c) magnesium and dilute hydrochloric acid?
(d) magnesium and dilute sulfuric acid?
2. What causes the observations recorded in Step 3 of the experiments? Support with balanced chemical equations.
3. What happened in Steps 2 and 3 when lead and copper were used in the experiment?Write the balanced chemical equations for the reactions of these metals with sulfuric acid.
Task 2
Use virtual laboratories or interactive simulations and animations to explore the strength and the basicity of acids.
STRENGTHS OF ACIDS
The strength of an acid is a measure of its ability to ionise (dissociate) in water to produce hydrogen ions (H⁺). The more the acid ionises in water, the stronger it is. The strength of the acid solution can be determined by its pH value. The pH is the measure of the degree of acidity or alkalinity of the solution. Based on their strengths, acids are grouped into strong acids and weak acids.
Strong acids ionise completely in water to give large amounts of H+. This complete ionisation makes them highly reactive. Common examples of strong acids are mineral acids such as hydrochloric acid, nitric acid, and sulfuric acid. For example, hydrochloric acid ionises completely in water to give H⁺ and Cl⁻.
HCl(aq)⟶H+(aq)+Cl−(aq)
Weak acids ionise partially in water to produce small amounts of H+. This means that not all acid molecules release H+. Common examples of weak acids are ethanoic acid, carbonic acid, and citric acid. For example, ethanoic acid (CH₃COOH) ionises partially in water to produce few CH₃COO⁻ and H⁺.
CH3COOH(aq)↽−−⇀CH3COO−(aq)+H+(aq)
Note:
(a) The pH values of solutions increase with decrease in acidity; therefore, weak acids have higher pH values than strong acids.
(b) Solutions of strong acids are good conductors of electricity because they contain more free mobile ions to carry the charges than solutions of weak acids.
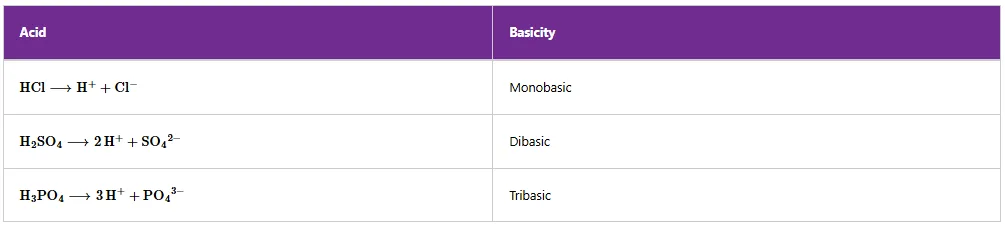
BASICITY OF AN ACID
Basicity of an acid is the number of ionisable hydrogen atoms per molecule of the acid that can be displaced by a metal in solution. For example, hydrochloric acid is monobasic as it has one ionisable hydrogen atom that can be displaced. Sulfuric acid and phosphoric acid are dibasic and tribasic, respectively. Sulfuric acid has two ionisable hydrogen atoms, while phosphoric acid has three ionisable hydrogen atoms that can be displaced (Table 5.2).
Table 2.Basicity of some acids
Exercise 1
1. Describe how the degree of ionisation leads to differences in acidic strength.
2. Should the strength of an acid be the only factor in determining its use in industry, home or laboratory? Explain your answer.
3. A particular skincare product contains glycolic acid. Why is it important to know the strength of the acid before using it on the skin?
4. Why do the physical properties of acids make them suitable for cleaning products?
BASES
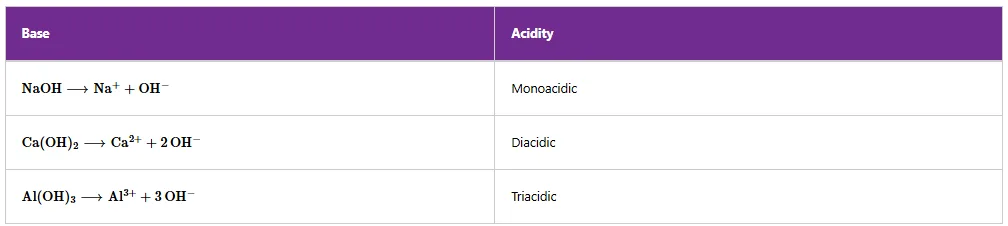
A base is a substance that neutralises an acid by reacting with hydrogen ions. Bases include the oxides, hydroxides, and carbonates of metals.
Metal oxides are those compounds which contain a metal and an oxygen atom. Examples of metal oxides include sodium oxide, Na₂O; potassium oxide, K₂O; calcium oxide, CaO; magnesium oxide, MgO; and copper(II) oxide, CuO.
Hydroxides are those compounds which contain a hydroxy group (-OH). Examples of hydroxides include calcium hydroxide, Ca(OH)₂; magnesium hydroxide, Mg(OH)₂; sodium hydroxide, NaOH; potassium hydroxide, KOH; and ammonium hydroxide, NH₄OH.
Metal carbonates are made up of a metal ion and a carbonate ion. Examples of metal carbonates include sodium carbonate, Na₂CO₃; potassium carbonate, K₂CO₃; magnesium carbonate, MgCO₃; and calcium carbonate, CaCO₃.
Bases are abundant in nature. Examples of natural substances that contain bases are ashes, banana peels, and avocado. In addition, other bases occur naturally on land and in water bodies; such bases include soda ash (sodium carbonate), baking soda (sodium bicarbonate) and limestone (calcium carbonate) Figure 5.6 shows some sources of natural bases.


Some sources of natural bases
Many common household products such as detergents, deodorants, toothpaste, and baking powder contain bases. These products contain synthesised bases.
Some bases are readily soluble in water, while others are not. Soluble bases are called alkalis. An alkali is a soluble base which, when dissolved in water, forms hydroxide ions. Therefore, all alkalis are bases, but not all bases are alkalis. Examples of soluble and insoluble bases are shown in Table 5.3.
Table 3: Examples of soluble and insoluble bases
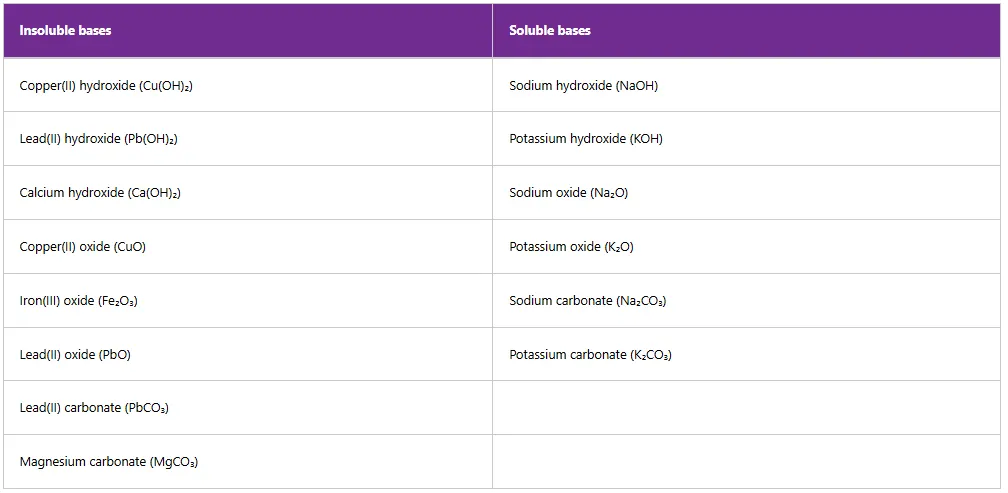
Note: Calcium hydroxide and lead hydroxide are moderately soluble in water. Oxides and hydroxides of other metals are insoluble except those of Group I elements.
PROPERTIES OF BASES
Bases have physical and chemical properties that differ from those of acids.
A. Physical properties of bases
Bases have the following physical properties:
1. Most bases have a bitter taste. For example, milk of magnesia, a common antacid.
2. Bases have a ’soapy’ or slippery feel. For example, a rub of a drop of baking powder solution between fingers gives a slippery feel.
3. Most bases are insoluble in water.
4. Bases generally do not have odour except for ammonia which has a pungent smell.
5. Bases are corrosive depending on their pH and concentrations.
6. Soluble bases (alkalis) conduct electricity when dissociated into ions.
B. Chemical properties of bases
Bases have the following chemical properties:
1. The pH values of bases are greater than 7.
2. Bases turn red litmus paper blue as shown in Figure 5.7.

Red litmus paper immersed in an alkali solution
3. Bases change the colour of phenolphthalein (POP) indicator pink and that of methyl orange (MO) indicator yellow.
4. Soluble bases react with most cations to precipitate hydroxides. In this case, they precipitate insoluble metal hydroxides from their salt solutions. For example, potassium hydroxide reacts with iron(III) chloride to yield iron(III) hydroxide and potassium chloride.
3KOH(aq)+FeCl3(aq)⟶Fe(OH)3(s)+3KCl(aq)
Ionically, the reaction is represented as:
3OH−(aq)+Fe3+(aq)⟶Fe(OH)3(s)
5. Bases react with acids to form salt and water. For example, sodium hydroxide reacts with dilute hydrochloric acid to produce sodium chloride and water.
NaOH(aq)+HCl(aq)⟶NaCl(aq)+H2O(l)
The ionic equation is
OH−(aq)+H+(aq)⟶H2O(l)
6. Alkalis react with ammonium salts to produce ammonia gas.
KOH(aq)+NH4Cl(s)⟶KCl(aq)+H2O(l)+NH3(g)
STRENGTHS OF BASES
Activity 2
Aim: To identify acidic and basic substances
Requirements: Coconut, citrus fruits, ripe tomatoes, vinegar, sour milk, wood ash, pieces of chalk, yoghurt, blender, knife, water, red litmus papers, blue litmus papers, and beakers
Procedure
1. Prepare juices of coconut, citrus fruits, and ripe tomatoes.
2. Use litmus papers to test the acidity or basic nature of the juices obtained in Step 1.
3. Mix some wood ash and chalk powder, each with little water, decant to obtain a supernatant and use litmus papers to test the acidity or basic nature of the supernatant.
4. Take small amounts of vinegar, sour milk, and yoghurt into separate beakers and test for acidity or basic nature using litmus papers.
Questions
1. Which substances are acidic and which ones are basic?
2. How relevant is this activity to your daily life?
NEUTRALISATION REACTION
If an acid and a base are mixed together in correct amounts, a neutral solution is produced. A reaction of this type is called neutralisation. Neutralisation is therefore, a reaction between an acid and a base to produce salt and water.
It is essentially the reaction between the hydroxide ions found in the basic solution and the hydrogen ions found in an acidic solution. The reaction is referred to as neutralisation because the resulting products are neither basic nor acidic.
Acid+Base⟶Salt+Water
Neutralisation reactions that involve the reaction of an acid with a carbonate or hydrogencarbonate produce carbon dioxide gas in addition to salt and water.
Acid+Carbonate/hydrogencarbonate⟶Salt+Water+Carbon dioxide
The following are examples of neutralisation reactions between bases and dilute acids:
Nitric acid reacts with potassium hydroxide to yield potassium nitrate and water.
HNO3(aq)+KOH(aq)⟶KNO3(aq)+H2O(l)
Sulfuric acid reacts with calcium hydroxide to yield calcium sulfate and water
H2SO4(aq)+Ca(OH)2(aq)⟶CaSO4(s)+2H2O(l)
Hydrochloric acid reacts with sodium carbonate to yield sodium chloride, water, and carbon dioxide gas.
2HCl(aq)+Na2CO3(aq)⟶2NaCl(aq)+H2O(l)+CO2(g)
Hydrochloric acid reacts with sodium hydrogencarbonate to yield sodium chloride, water, and carbon dioxide gas.
HCl(aq)+NaHCO3(aq)⟶NaCl(aq)+H2O(l)+CO2(g)
To identify the end point for an acid-base reaction, an indicator must be added. For example, when phenolphthalein indicator is used in a particular titration (by running the acid from a burette to a base in a flask), the pink colour of the content in the flask starts to change slowly to colourless.
The end point is marked when the reaction mixture in the flask just turns colourless. During neutralisation of a base with an acid, the volume of the base in a flask is normally known. The volume of the acid that has neutralised the alkali is called a titre value or titre volume.
To get the exact volume (V) of the acid that neutralises a base, the initial burette reading, V1 is subtracted from the final burette reading V2.
V = V2 – V1
Activity 3
Aim: To investigate the neutralisation reaction between sodium hydroxide and dilute hydrochloric acid
Requirements: Pipette, burette, retort stand and clamp, 10-mL measuring cylinder, conical flasks, beakers, evaporating dish, white tile, heat source, tripod stand, wire gauze, 0.1 M sodium hydroxide, 0.1 M hydrochloric acid, and phenolphthalein indicator
Procedure
1. Transfer 25 cm³ (or 20 cm³) of sodium hydroxide solution into a conical flask by using a pipette.
2. Add 2 to 3 drops of phenolphthalein indicator to the solution.
3. Set the apparatus as shown in Figure 5.8.

Set-up of the acid-base titration
4. Fill the burette with dilute hydrochloric acid and record the initial volume, V1.
5. Run the acid into the alkali (base), a few drops at a time, until the indicator just changes colour. Swirl the components of the flask after every addition of the acid.
6. Record the new level of the remaining acid in the burette as V2. Calculate the volume of the acid used in the reaction.
7. In a clean beaker, mix the same volume of the base used with the same volume of the acid obtained from the titration. Do not add any indicator.
8. Transfer 10 cm³ of the resulting solution into an evaporating dish and carefully heat the solution until crystals start to form.
9. Stop heating and allow the remaining liquid to evaporate, leaving behind a white solid.
Questions
1. Why is the phenolphthalein indicator important in an acid-base experiment?
2. What is the balanced chemical equation representing the reaction in the experiment? Give the name of the white solids obtained in Step 9.
3. What is the ionic equation of the reaction in the experiment?
Applications of neutralisation reactions
Neutralisation reactions have many applications in daily life. Some of the applications are explained as follows:
Treating insect stings and bites
Insect bites or stings, and plants with stinging hair often cause small and red lumps on the skin which may be painful and itchy. Insects such as bees, have stings that inject an acidic liquid in the blood through the skin.
The stings can be neutralised by rubbing baking soda on the affected area. Ant bites and nettle (a plant with stinging hair) stings contain methanoic acid which is neutralised by using baking soda or other alkaline substances such as cucumber and avocado. Wasp stings are alkaline and can be neutralised with vinegar which contains acetic acid. Figure 5.9 shows different sources of stings or bites.



Relieving indigestion
Indigestion is a discomfort in the stomach that is associated with difficulty in digesting food. It is caused by the presence of excess acid like hydrochloric acid in the stomach. The excess acid can be neutralised by taking a liquid or tablets that contain magnesium or sodium hydrogencarbonate commonly known as antacids.
Soil treatment
Most plants grow well in soils that have optimal pH values. When soils are too acidic, for example, due to applications of ammonium based fertilisers, chemicals such as calcium oxide (quicklime) and calcium hydroxide (slaked lime) are added to adjust the soil pH (Figure below). Such chemicals that neutralise the soil acidity are called liming materials.


Soil liming
Treating factory wastes
Liquid wastes from factories often contain acids and bases. If the wastes get into water bodies such as lakes, ponds and rivers, they can harm aquatic organisms like fish. Acidic wastes can be controlled by adding bases such as calcium hydroxide to neutralise them.
Preventing formation of acid rain
When the pH of rainwater falls below 5.6, it is called acid rain. Acid rain is caused by chemical reactions between rainwater and gases such as sulfur dioxide and nitrogen dioxide which are released into the atmosphere. Acid rain increases the acidity of soils, rivers and lakes and adversely affects vegetation and aquatic organisms. To reduce this problem, air pollution devices containing bases are fitted in exhaust pipes and chimneys to neutralise the acidic compounds before reaching the atmosphere.
Neutralising accidental spills
If an acid or an alkali spills on the floor or work surface in the laboratory, it can be neutralised. For example, sulfuric acid which is very corrosive, can be neutralised by adding sodium hydroxide.
Manufacturing fertilisers
The production of ammonium fertilisers is done through the neutralisation of ammonia with a mineral acid. Ammonium nitrate for example, is produced by the reaction of ammonia with nitric acid.
NH3(g)+HNO3(aq)⟶NH4NO3(aq)
Ammonia gas also reacts with sulfuric acid to give ammonium sulfate (NH₄)₂SO₄ fertilizer (Figure below).
2NH3(g)+H2SO4(aq)⟶(NH4)2SO4(aq)
Sack of ammonium sulfate fertiliser

USES OF ACIDS AND BASES
Task 2
Explore reliable books, online, or home resources to identify three everyday products containing acids and three containing bases.
For each product or material you find:
1. Write the trade name, common name and chemical name of the product.
2. Identify the acid or base it contains (if known).
3. Describe its use or purpose in daily life.
Acids and bases have many uses in daily life
For instance, some plants grow well in acidic soil, but others grow better in a neutral or alkaline soil. Farmers need to know the pH of the soil because the soil can be too acidic causing inability of plants to grow well and reduce the yields. Bases can be used by farmers to reduce the acidity of the soil, and acids can be used to correct the pH for the basic soils.
Amino acids and some vitamins such as vitamin C are important for our health. Sulfuric acid is used in car batteries. Citric acid is used in food preservation. Bases such as sodium hypochlorite are used in bleaching to remove stains on clothes.
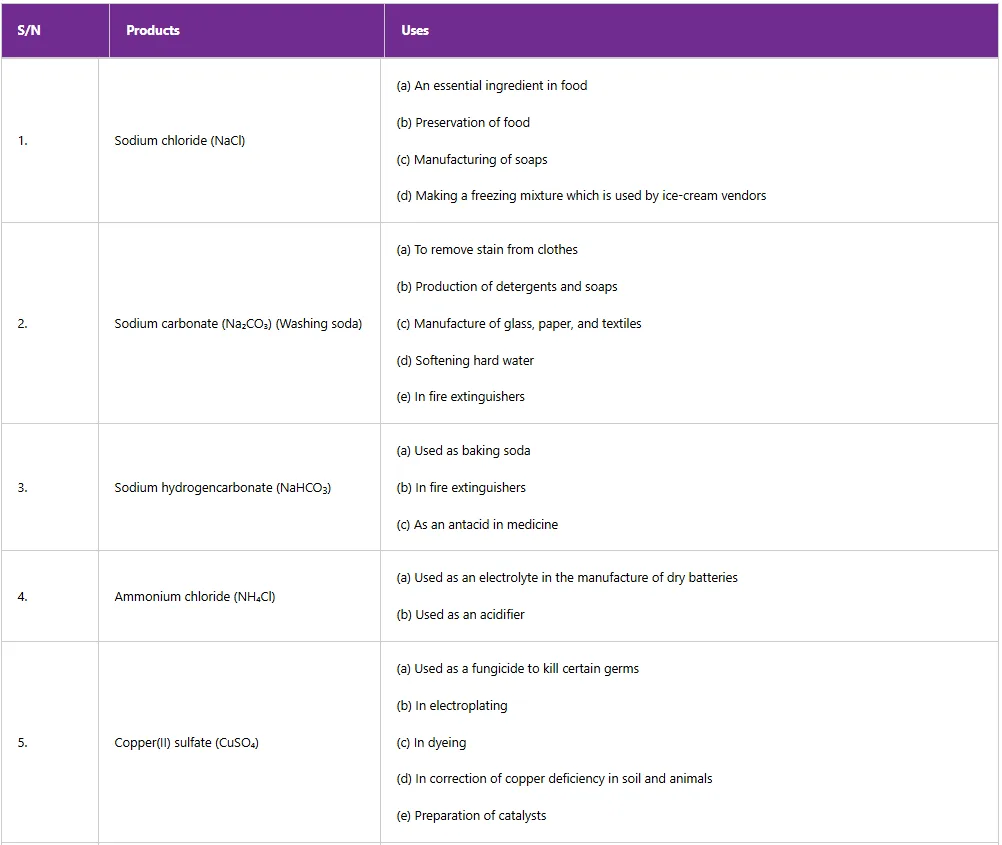
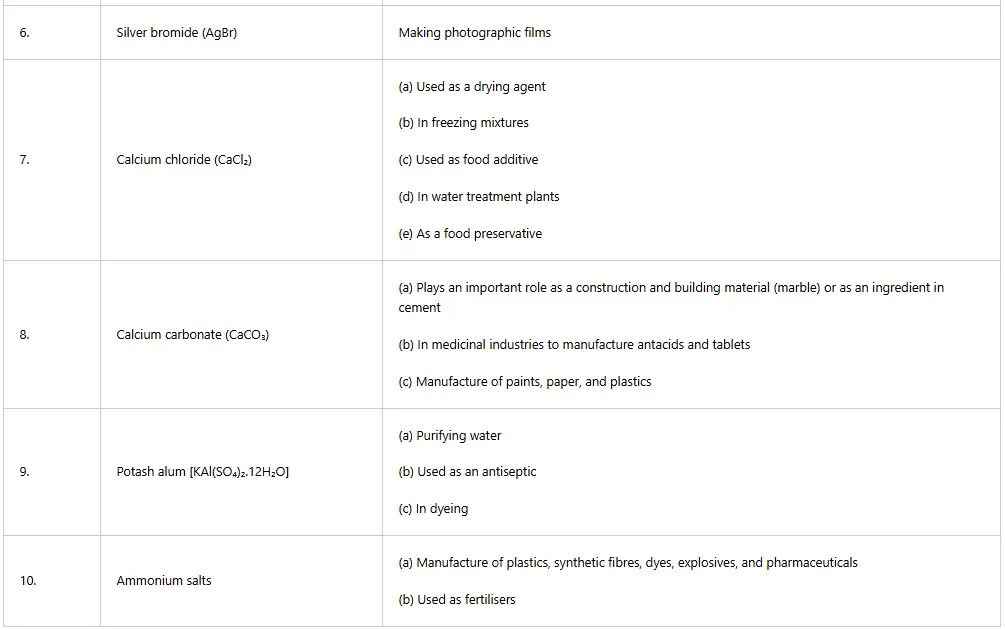
Ammonium hydroxide is used as a glass cleaner, and magnesium hydroxide is used as a laxative to regulate bowel movements. Magnesium hydroxide is also used as an antacid to relieve indigestion, sour stomach and heartburn. Tables 5.5 and 5.6 summarise the uses of some acids, and bases, respectively.
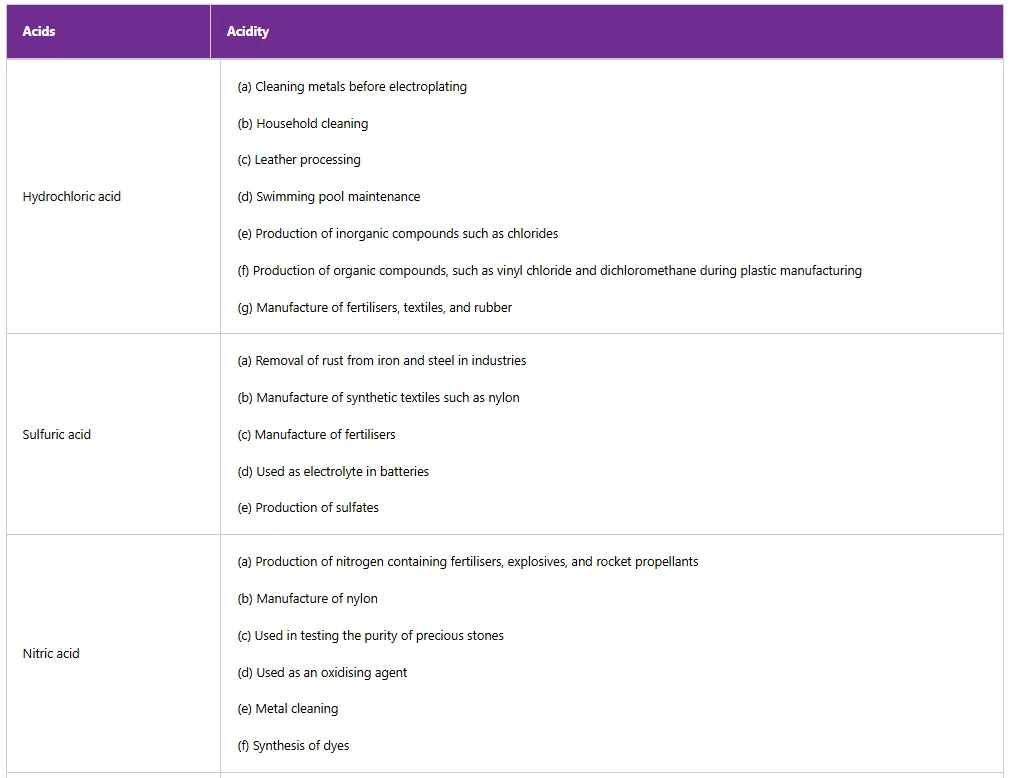
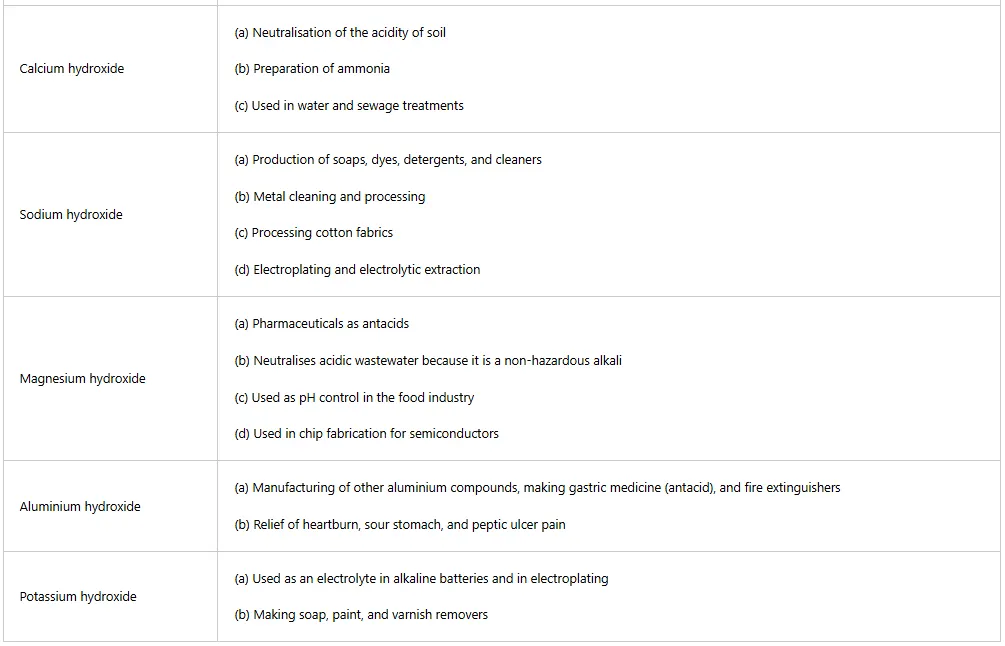
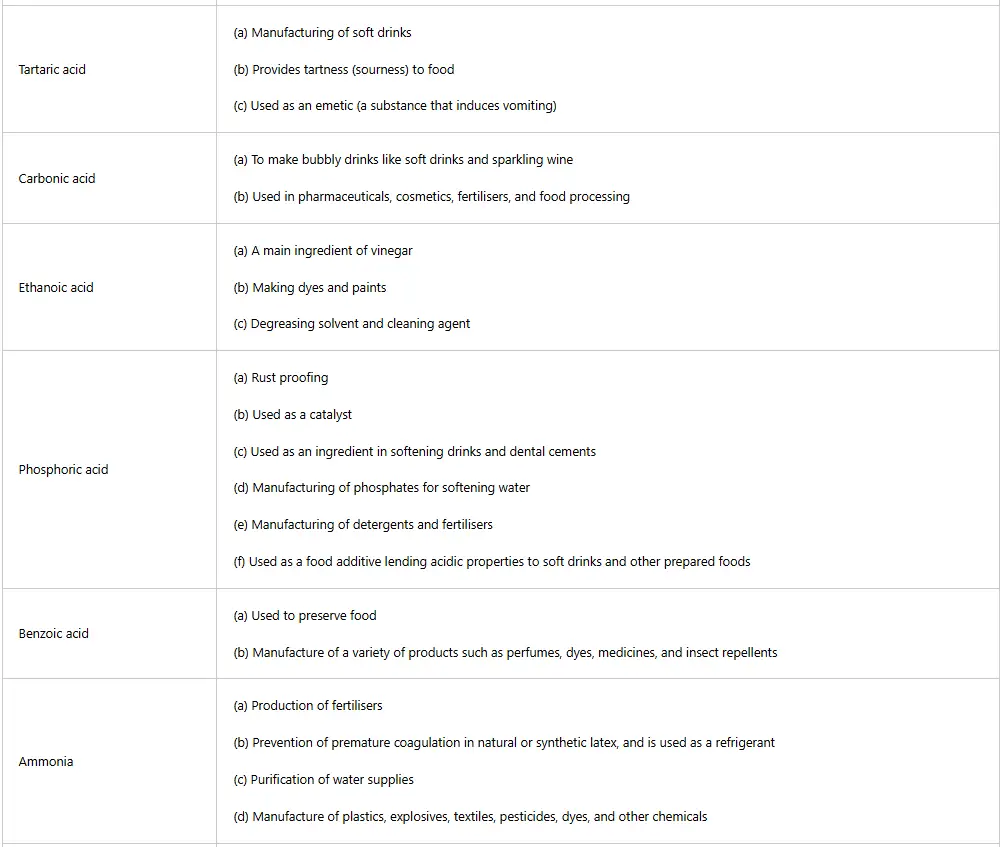
Table 5: Uses of some acids in daily life
Exercise 2
1. Group the following substances into either acids, bases or neutral solutions: Ash solution, fresh milk, sour milk, liquid soap, chalk solution, tomato juice, lemon juice, sugar solution, table salt solution, and cucumber juice.
2. Strong bases, such as sodium hydroxide, are used in industrial cleaning, while weak bases, such as baking soda, are safe for home uses. Why is this difference important in terms of safety and effectiveness?
3. A bee stung a gardener while working in the garden. What household material can be used to relieve the pain in the stung area? Explain your answer.
ACID-BASE INDICATORS
Acid-base indicators are chemicals that are used to determine the chemical nature of a substance; whether it is acidic or basic.
The acid-base indicators are also known as pH indicators because acidity and alkalinity relate to pH range.
Indicators normally change colour to indicate the presence or absence of acids or bases. The pH indicators operate efficiently over a certain pH range.
pH SCALE
pH scale is a scale of numbers, from 0 to 14 which is used to express acidity, neutrality, and alkalinity. The pH 7 indicates neutrality of a substance. Any substance that has a pH below 7 is acidic, while a substance with pH above 7 is basic.
The smaller the number below 7 in the scale, the more acidic is the substance. Similarly, the greater the number above 7, the more basic is the substance.
This means that an acid with pH 1 is more acidic than that with pH 6 and the base with pH 14 is more basic than that with pH 8. Figure 5.12 shows the trends in acidity and alkalinity in the pH scale.

Trends in acidity and alkalinity
OCCURRENCE OF PH INDICATORS
pH indicators can occur naturally or be synthesised.
Natural pH indicators
A natural pH indicator is a substance which is found naturally and can be used to determine whether the substance is acidic or basic. Examples of natural pH indicators are turmeric, grape juice, red cabbage, cassava leaves, onion, beetroot, and coloured flowers like that of rosella and hibiscus.
For example, red cabbage juice is pink in acids and green in bases. An extract of hibiscus flowers has a red colour in an acid and greenish yellow colour in a base. Onion is one of the olfactory indicators which diminishes its smell in a base and remains as it is in an acid. Vanilla essence has similar characteristics to onion. The smell of vanilla essence disappears when it is added to a base, but persists when it is added to an acid.
The most commonly used natural indicator is litmus which is extracted from lichens (Figure below). Litmus has purple, red, and blue colours in distilled water, acidic solution, and basic solution, respectively. Litmus is available either in solution, or in the form of strips of paper.

Lichen
Activity 4
Aim: To prepare a pH indicator from hibiscus flowers
Requirements: Hibiscus flowers, water, beakers, droppers, mortar and pestle, knife, 50% ethanol solution, heat source, evaporating dish, dilute hydrochloric acid, and dilute sodium hydroxide
Procedure
1. Cut the hibiscus flowers into small pieces and then crush them by using mortar and pestle.
2. Add about 20 cm³ of ethanol solution into the crushed hibiscus flowers and continue crushing. Decant the mixture obtained and then keep the solution obtained aside.
3. Boil the solution obtained to make sure that all the ethanol evaporates. Leave the residue to cool.
4. Add about 20 cm³ of distilled water to the residue and stir to obtain an indicator solution.
5. Establish the colour of an indicator prepared by testing it with hydrochloric acid and sodium hydroxide, one at a time.
Questions
1. How can the colour changes observed with hibiscus flower extract help determine whether a substance is an acid or a base?
2. Why was ethanol used as a solvent in the extraction instead of water?
3. What other substances found in your environment can be used as acid-base indicators?
Activity 5
Aim: To prepare a pH indicator from cassava leaves
Requirements: Fresh cassava leaves, distilled water, ethanol, mortar and pestle or blender, filter paper, test tubes or small clear containers, dropper or pipette, known acidic and basic solutions (for example, vinegar, lemon juice, baking soda solution, soap solution), pH paper or a pH meter
Procedure
1. Collect fresh cassava leaves and rinse them thoroughly with distilled water to remove any dirt or impurities.
2. Chop the leaves into small pieces and then grind them thoroughly using a mortar and pestle. Alternatively, use a blender to blend the leaves into a fine pulp.
3. Add a small amount of distilled water to facilitate the grinding or blending process.
4. Add a small volume of ethanol to the mixture to enhance the extraction of pigments, especially anthocyanins.
5. Filter the homogenous mixture using filter paper to separate the liquid extract from the solid leaf residues. Collect the filtrate in a clean beaker.
6. Establish the colour of an indicator prepared by testing it with the acidic and basic solutions available, one at a time.
Questions
1. What colour changes were observed using cassava leaf extract indicator in the substances tested?
2. How can the knowledge gained from this activity be used in testing spoiled fruits?
Synthetic pH indicators
Synthetic pH indicators are chemical substances which are made from different substances in the laboratories used to determine the pH of substances. Examples of synthetic indicators include universal indicator, phenolphthalein (POP), and methyl orange (MO).
Universal indicator
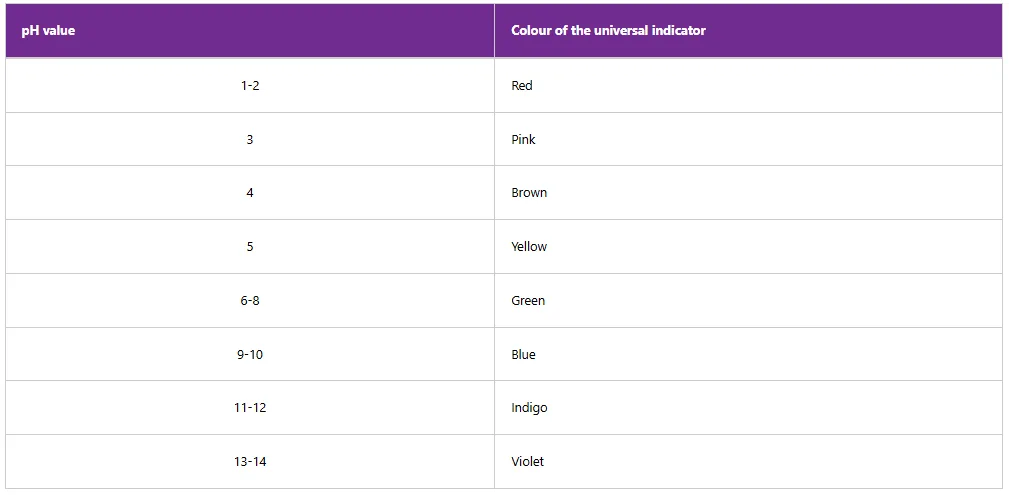
The universal indicator is a chemical substance that displays several colour changes across a wide range of pH values as shown in Table 5.7. Basically, the universal indicator is a blend of pH indicator solutions which include phenolphthalein, methyl red, bromothymol blue, and thymol blue.
When an acid or base solution is added to the universal indicator, a new colour is produced, which is used to determine the pH value of the acid or the base solution by matching the colour with the colours on the pH colour chart.
Table 7: pH ranges of the universal indicator
Other indicators
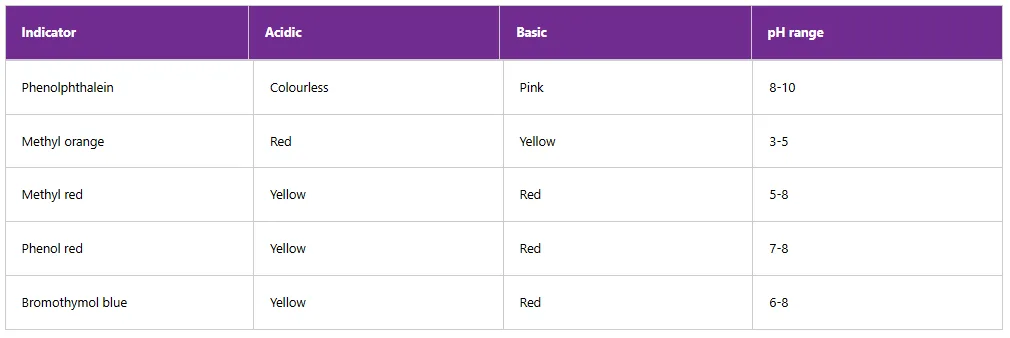
The indicators such as methyl orange, methyl red, bromothymol blue, phenol red, thymol blue and phenolphthalein are used to test the acidity or alkalinity of acids and bases. Table 5.8 shows the colour changes and the pH ranges of selected indicators.
Table 8: Colour changes of common indicators in acidic and basic solutions
SALTS
Task 5
Use online simulation and interactive tools or any other reliable resources to investigate the properties, types, and everyday applications of salts.
A salt is an ionic compound which is made up of positively charged ions (cations) and negatively charged ions (anions). Salts occur naturally and are also synthesised in the laboratory. The main natural sources of salts include sea water and rocks.
Different salts are used in our homes and industries for various purposes. These include table salt (NaCl), washing soda (Na₂CO₃.10H₂O), Plaster of Paris (POP) (CaSO₄.H₂O), and bleaching powder (Ca(OCl)₂).
TYPES OF SALTS
Salts are categorised into neutral salts, acidic salts, and basic salts depending on their pH values when dissolved in water. Salts can also be categorised as normal salts, and double salts depending on their formation and compositions.
Neutral salts
These are formed by the reactions of strong acids and strong bases. They are called neutral salts because their aqueous solutions are neutral to litmus. Neutral salts, when categorised based on formation and composition, are termed as normal salts.
A normal salt is formed by a complete replacement of replaceable hydrogen atoms from an acid molecule by a metal. Examples of these salts are NaCl, KCl, K₂SO₄, KClO₃, MgCl₂, CuSO₄, MgSO₄, and NaNO₃.
Acidic salts
Acidic salts are those salts that produce acidic solutions when dissolved in water. They are formed when a strong acid reacts with a weak base. Their aqueous solutions turn blue litmus paper red.
Acidic salts may contain one or more replaceable hydrogen atom(s) present in an acid molecule or positive radical. Examples of acidic salts include FeCl₃, ZnCl₂, HgCl₂, Fe₂(SO₄)₃, NaHSO₄, NaHPO₄, (NH₄)₂SO₄, and NH₄Cl.
Basic salts
Basic salts are a class of salts that produce alkaline solutions when dissolved in water. Their aqueous solutions turn red litmus paper blue. A basic salt is a product of a reaction between a strong base and a weak acid. Examples of basic salts include CH₃COONa, Na₂CO₃, NaHCO₃, and Na₂C₂O₄.
Double salts
A double salt is a mixture of two salts, which dissolves in water to give two different cations or anions. Examples are potash alum [KAl(SO₄)₂.12H₂O], ferric alum [NH₄Fe(SO₄)₂.12H₂O], and Mohr’s salt [(NH₄)₂Fe(SO₄)₂.6H₂O].
PROPERTIES OF SALTS
There are several methods of preparing salts. The selection of the method to be used depends on whether the salt required is soluble or insoluble in water.
Preparation of soluble salts
Soluble salts are prepared by reacting acids with metals, alkalis, insoluble bases or carbonates.
Reactions of acids with metals (displacement method)
The displacement method is suitable for preparing salts of moderately reactive metals such as zinc, iron, lead, tin, and copper. Highly reactive metals such as K, Na, and Ca react very rapidly with acids and can cause explosions. The displacement reaction occurs as shown in the general equation:
Metal+Acid⟶Salt+Hydrogen gas
For example, zinc displaces hydrogen from sulfuric acid as shown in the following reaction equation:
Zn(s)+H2SO4(aq)⟶ZnSO4(aq)+H2(g)
Activity 7
Aim: To prepare zinc sulfate
Requirements: Beaker, evaporating dish, glass rod, spatula, tripod stand, wire gauze, filter papers, filter funnel, heat source, zinc granules, and 1 M dilute sulfuric acid
Procedure
1. Put about 25 cm³ of 1 M dilute sulfuric acid in a beaker, and then add some zinc granules into it. Mix the contents by using a glass rod.
2. Continue adding more zinc to the beaker to excess until no more effervescence is observed.
3. Filter out the excess zinc and pour the filtrate into an evaporating dish.
4. Heat the filtrate while ensuring that not all the water is evaporated.
5. Allow the heated filtrate to cool and then filter off any excess liquid.
6. Allow the crystals to dry by rubbing them between filter papers. Record the observations.
Questions
1. Why is excess zinc used in this experiment?
2. Why is it not allowed to dry up completely the filtrate when evaporating?
3. What will happen if magnesium ribbon is used instead of zinc granules. Support your answer with a chemical equation.
Reactions of acids with metal oxides and metal hydroxide (neutralisation method)
The neutralisation method is usually used to prepare soluble salts such as sodium, potassium, and ammonium salts. Metal oxides and metal hydroxides react with acids to produce salt and water only.
Metal oxide+Acid⟶Salt+Water
Metal hydroxide+Acid⟶Salt+Water
Activity 8
Aim: To prepare potassium chloride
Requirements: Pipette, burette, conical flasks, dropper, evaporating dish, tripod stand, wire gauze, heat source, 2 M potassium hydroxide solution, 2 M hydrochloric acid solution, phenolphthalein indicator, and distilled water
Procedure
1. Transfer 25 cm³ of 2 M potassium hydroxide solution into a conical flask.
2. Add two drops of phenolphthalein indicator.
3. Fill the burette with 2 M of hydrochloric acid. Record the initial reading.
4. Slowly run the acid from the burette into the conical flask while gently swirling the flask to mix the acid and the base. Close the burette when the solution just changes its colour. Record the volume of the acid used.
5. Transfer the mixture obtained into an evaporating dish and heat until all the water evaporates.
Questions
1. What was the colour change of the reaction?
2. What volume of acid was used in the entire reaction?
3. What happened in Step 4? Explain with the aid of a chemical equation.
Reactions of acids with insoluble bases
Reactions of acids with insoluble bases result in the formation of soluble salts. For example, insoluble bases of zinc oxide and copper(II) oxide react with acids to form soluble salts like zinc sulfate, copper(II) sulfate, copper(II) chloride, and water.
Insoluble base+Acid⟶Salt+Water
ZnO(s)+H2SO4(aq)⟶ZnSO4(aq)+H2O
Activity 9
Aim:To prepare copper(II) chloride
Requirements: 100-mL measuring cylinder, tripod stand, 250-mL beaker, glass rod, evaporating dish, wire gauze, filter papers, funnel, heat source, 2 M hydrochloric acid solution, and copper(II) oxide,
Procedure
1. Pour about 50 cm³ of 2 M hydrochloric acid into a beaker and heat gently.
2. Dissolve some copper(II) oxide little by little in the hot acid and stir for some time until the metal oxide is in excess.
3. Filter off the solid and heat the filtrate until it approaches to dryness.
Questions
1. What chemical reaction took place in this experiment? Explain with a balanced chemical equation.
2. Why was it necessary to use hot hydrochloric acid in the reaction with copper(II) oxide?
3. Why did you heat the fitrate?
4. What is the colour of the product formed in Step 3?
Reactions of acids with metal carbonates
Most metal carbonates such as calcium carbonate, copper(II) carbonate and potassium carbonate react with acids to produce salt, water and carbon dioxide gas as shown in the following equation:
Carbonate+Acid⟶Salt+Water+Carbon dioxide
Activity 10
Aim: To prepare copper(II) sulfate
Requirements: Beaker, evaporating dish, glass rod, wire gauze, measuring cylinder, filter paper, filter funnel, tripod stand, heat source, dilute sulfuric acid, and copper(II) carbonate
Procedure
1. Transfer about 25 cm³ of dilute sulfuric acid into a small beaker and heat gently.
2. Add solid copper(II) carbonate to the beaker containing the hot acid and stir the mixture by using a glass rod until it is saturated.
3. Filter the mixture and crystallise the filtrate by heating gently until almost all of the water evaporates. Do not allow evaporation to dryness.
Questions
1. Which reaction occurred in this experiment? Explain with a balanced chemical equation.
2. Why was the mixture filtered before evaporation?
3. How can the gas formed in this experiment be tested?
Reactions of acids with metal hydrogencarbonates
Metal hydrogencarbonates, or commonly known as bicarbonates, are compounds in the form of MHCO3, where M represents a metal. These compounds react with acids in the same way as metal carbonates to form a salt, water, and carbon dioxide gas. For example, sodium hydrogencarbonate (NaHCO3) reacts with dilute hydrochloric acid to produce sodium chloride, water, and carbon dioxide gas.
HCl(aq)+NaHCO3(aq)⟶NaCl(aq)+CO2(g)+H2O(l)
Preparation of salts by direct combination
If salts are required in anhydrous forms, they cannot be prepared by wet methods. In this case, it is possible to prepare them by direct combination. For example, in the preparation of iron(III) chloride, dry chlorine gas is passed over hot iron wire or steel wool as shown in Figure 5.14. The iron(III) chloride vapour produced is condensed to give a solid salt. This is indicated in the following equation:
2Fe(s)+3Cl2(g)→heat2FeCl3(s)

Set-up of the direct preparation of iron(III) chloride
Preparation of insoluble salts
Insoluble salts are prepared by using ionic precipitation or a double decomposition method. In this method, two soluble salts are used to form an insoluble salt and a soluble salt. The insoluble salt precipitates, while the soluble salt remains in solution.
The precipitate is filtered and washed with distilled water and then dried. For example, to prepare insoluble lead(II) iodide, the solution containing lead ions and another containing iodide ions are mixed. Such solutions could be lead(II) nitrate and potassium iodide. A bright yellow precipitate of lead(II) iodide is formed after mixing. The chemical equation for this reaction is as follows:
Pb(NO3)2(aq)+2KI(aq)⟶PbI2(s)+2KNO3(aq)
Other common insoluble salts which can be prepared by using this method include calcium sulfate (CaSO₄), magnesium carbonate (MgCO₃), silver chloride (AgCl), barium carbonate (BaCO₃), barium sulfate (BaSO₄), and lead(II) sulfate (PbSO₄).
Task 6
Use online search tools or other reliable resources to investigate and analyse various methods used in salt production in Tanzania.
PROPERTIES OF SALTS
Salts exhibit different physical and chemical properties.
A. Physical properties of salts
The physical properties of salts include their appearance in colour, texture, and solubility.
Physical appearance of salts
Salts appear in crystalline or powder forms and exhibit different colours depending on their types. For example, hydrated copper(II) sulfate is a blue crystalline salt, while sodium chloride is a white crystalline salt.
Ferrous chloride is a green crystalline salt, while calcium nitrate is a white crystalline salt. Calcium carbonate is a white powder. Figure 5.15 shows some examples of crystalline and powdered salts.





Crystalline and powdered salts
Solubility of salts
Solubility of a solute in a given solvent is the number of the grams of the solute required to saturate 100 grams of solvent at a given temperature, or it is the amount of grams or moles of a substance that dissolve in a solvent to form a saturated solution at a given temperature.
The solubility of a solute in a solvent is not fixed but varies with temperature. Some solutes are more soluble in certain solvents than others. For example, 40 g of sodium chloride can dissolve in 100 g of water at 50 °C, while 80 g of potassium nitrate can dissolve in 100 g of water at the same temperature.
In this case, potassium nitrate is more soluble than sodium chloride. Some salts such as sodium chloride and potassium nitrate are soluble in water, while others such as calcium carbonate and lead chloride are insoluble.
The solubility behaviour of some salts is summarised in Table below.
Table 9: Soluble and insoluble salts

Activity 11
Aim: To investigate the solubility of salts
Requirements: Ten test tubes, test tube racks, 10-mL measuring cylinder, sodium carbonate, calcium nitrate, calcium carbonate, sodium sulfate, potassium chloride, barium sulfate, barium chloride, zinc chloride, lead chloride, ammonium carbonate, distilled water, and spatulas
Procedure
1. Put about 5 cm3 of distilled water into each test tube.
2. Put 0.2 g of each salt in each test tube containing an equal amount of water, then shake gently. Record the observations in each test tube.
Questions
1. Which salts dissolved readily?
2. Which salts dissolved slowly?
3. Which salts did not dissolve?
Chemical properties of salts
Different salts exhibit different chemical properties. For example, they decompose upon heating to produce different substances depending on the type of their metals and positions in the reactivity series.
Action of heat on salts
Different salts behave differently when heated. Some salts will decompose after slight heating, yet others have to be heated strongly to decompose. Some salts do not decompose at all, even with strong heating.
Action of heat on carbonates
Upon heating, metal carbonates decompose to form metal oxides and carbon dioxide gas. The decomposition reaction can be represented by the following equation, where M represents a particular metal:
MCO3(s)→heatMO(s)+CO2(g)
Sodium and potassium carbonates do not decompose even when strongly heated. The carbonates of magnesium and calcium decompose to give magnesium oxide and calcium oxide, respectively, with the evolution of carbon dioxide gas.
MgCO3(s)→heatMgO(s)+CO2(g)
CaCO3(s)→heatCaO(s)+CO2(g)
Other metal carbonates decompose easily on heating. For example, green copper(II) carbonate decomposes easily upon heating to give black copper(II) oxide and carbon dioxide gas.
CuCO3(s)→heatCuO(s)+CO2(g)
Zinc carbonate decomposes easily on heating to form zinc oxide and carbon dioxide gas. The zinc oxide produced is yellow when hot and white when cold.
ZnCO3(s)→heatZnO(s)+CO2(g)
Lead(II) carbonate decomposes when heated to give lead(II) oxide and carbon dioxide gas.
PbCO3(s)→heatPbO(s)+CO2(g)
Ammonium carbonate behaves so differently when heated. It decomposes slowly at room temperature to produce ammonia gas, water, and carbon dioxide.
(NH4)2CO3(s)→heat2NH3(g)+H2O(g)+CO2(g)
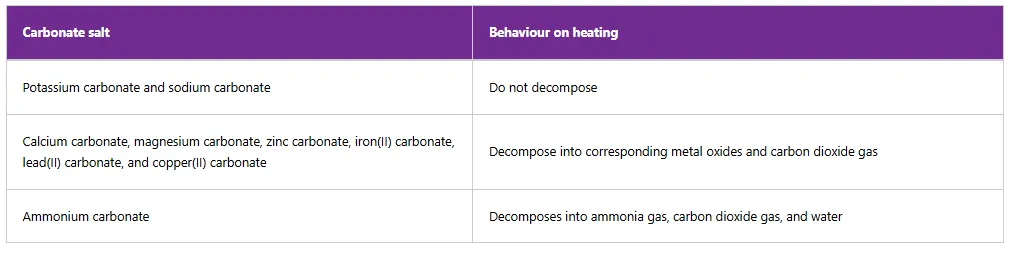
A strong pungent smell is produced due to the presence of ammonia gas. The evolved gas turns limewater milky, indicating the presence of carbon dioxide. It also turns a blue cobalt(II) chloride paper pink indicating the presence of water vapour. A red litmus paper turns blue due to the basic condition of ammonia gas in the solution. Table below summarises the behaviour of some carbonates on heating.
Table 10: Effects of heat on some carbonate salts
Activity 12
Aim: To investigate the effects of heat on carbonate salts
Requirements: Sodium carbonate, magnesium carbonate, calcium carbonate, copper(II) carbonate, zinc carbonate, lead(II) carbonate, limewater, test tubes, spatulas, heat source, blue litmus paper, wire gauze, tripod stand, test tube holder and bent delivery tube
Procedure
1. Put a spatulaful of zinc carbonate into a dry test tube.
2. Hold the test tube with a test tube holder and heat over a non-luminous flame.
3. Pass the gas evolved into a test tube of limewater. Record the results.
4. Put a moist blue litmus paper at the mouth of the test tube, and record the observations.
5. Observe the colour change of the compound when hot and when cold.
6. Repeat Steps 1–5 for the other carbonates. Record all the observations.
Questions
1. What differences were observed when heating zinc carbonate and sodium carbonate?
2. What is common when heating magnesium carbonate and copper(II) carbonate?
Activity 13
Aim: To investigate the effect of heat on ammonium carbonate
Requirements: Spatula, 2 test tubes, test tube holder, heat source, cobalt(II) chloride paper, red litmus paper, ammonium carbonate, and limewater
Procedure
1. Put a spatulaful of ammonium carbonate salt into a dry test tube.
2. Hold the test tube with a test tube holder and heat the salt over a non-luminous flame.
3. Pass the gas evolved into a test tube containing limewater. Record the observations.
4. Put a moist red litmus paper and cobalt(II) chloride paper at the mouth of the test tube, and record the observations.
Questions
1. What differences did you observe between this activity and Activity 5.12?
2. What is the balanced chemical equation for the reaction in Step 3?
Action of heat on nitrates
Most nitrates decompose on heating to give the metal oxide, brown fumes of nitrogen dioxide, and oxygen gas. Nitrates of alkali metals such as sodium and potassium decompose slowly on heating to give oxygen and metal nitrite.
2NaNO3(s)→heat2NaNO2(s)+O2(g)
Nitrates of calcium and copper decompose into their respective oxides, nitrogen dioxide gas, and oxygen gas.
2Ca(NO3)2(s)→heat2CaO(s)+4NO2(g)+O2(g)
2Cu(NO3)2(s)→heat2CuO(s)+4NO2(g)+O2(g)
Exceptional behaviours are shown by ammonium nitrate and silver nitrate. Ammonium nitrate decomposes to give dinitrogen oxide gas and water, whereas silver nitrate decomposes to give silver metal, nitrogen dioxide, and oxygen gas.
NH4NO3(s)→heatN2O(g)+H2O(l)
2AgNO3(s)→heat2Ag(s)+2NO2(g)+O2(g)
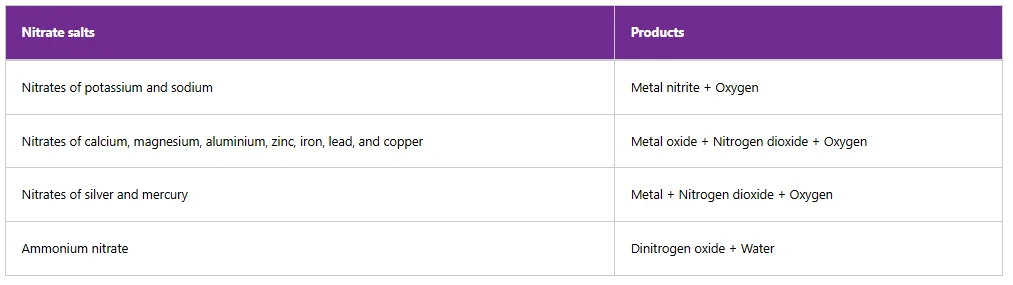
Table below summarises the effects of heat on nitrates.
Table 11: Effects of heat on some nitrate salts
Action of heat on sulfates
Sulfates are generally more stable to heat than nitrates. Even sulfates of metals that are low in the reactivity series must be strongly heated to decompose. Sulfates of alkali metals and alkaline earth metals do not decompose when heated. However, few sulfates such as ammonium sulfate and copper(II) sulfate decompose when heated. For example, ammonium sulfate decomposes on heating to give sulfuric acid and ammonia gas. During heating, the sulfate initially decomposes to give ammonium hydrogen sulfate as shown in the following equations:
(NH4)2SO4(s)→heatNH4HSO4(l)+NH3(g)
On cooling the products, ammonia gas, and sulfuric acid are formed.
NH4HSO4(l)→coolNH3(g)+H2SO4(l)
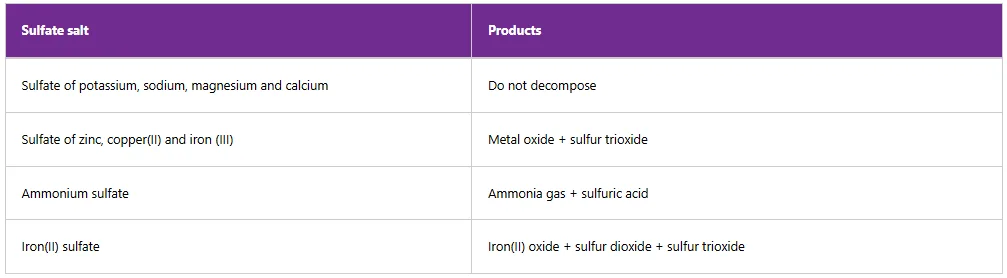
Table 5.12 summarises the effect of heat on some sulfate salts.
Table 12: Effect of heat on some sulfate salts
Activity 14
Aim: To investigate the action of heat on sodium sulfate and iron(II) sulfate
Requirements: Ignition tube, spatula, heat source, sodium sulfate, iron(II) sulfate, and red and blue litmus papers
Procedure
1. Put a spatulaful of sodium sulfate in the ignition tube and heat strongly.
2. Insert the wet blue and red litmus papers in the ignition tube while heating.
Record the observations.
3. Repeat Steps 1 and 2 by using iron(II) sulfate in the place of sodium sulfate.
Record the observations.
Questions
1. What were the colour changes in the two litmus papers?
2. What do the colour changes in the litmus papers imply?
Action of heat on chloride salts
All chloride salts except ammonium chloride do not decompose on heating. Ammonium chloride on heating, sublimes and decomposes to ammonia gas and hydrogen chloride gas.
NH4Cl(s)↽−−⇀heatHCl(g)+NH3(g)
Deliquescence, hygroscopy, and efflorescence in salts
Based on their behaviour when exposed to air, salts are categorised as deliquescent, hygroscopic or efflorescent.
Deliquescent salts
A salt that absorbs moisture from the air (environment) to form a solution is called a deliquescent salt. Examples of deliquescent salts are sodium hydroxide, phosphorus oxides, potassium hydroxide, and calcium chloride.
Hygroscopic salts
A hygroscopic salt is the one which absorbs water from air without necessarily forming a solution. Examples of hygroscopic salts are sodium chloride, sodium nitrate, calcium chloride, calcium oxide, and copper sulfate. The tendency of calcium chloride to absorb water vapour explains its use as a drying agent for many gases, except ammonia gas because it reacts with it. The water absorbed by the salt may result in changes in the physical properties, such as boiling point and viscosity.
Efflorescent salts
An efflorescent salt is the one which when left in air loses all the water of crystallisation. Examples of efflorescent salts are hydrated sodium carbonate (Na2CO3●10H2O), hydrated sodium sulfate (Na2SO4●10H2O), and hydrated iron(II) sulfate (FeSO4●7H2O).
USES OF SALTS
Salts have many uses in our daily lives. Some common uses include controlling soil pH, acting as antacids, serving as fertilisers, and helping to alleviate certain health disorders. They are also used for flavouring and preservating food, as well as in the production of pesticides.
Control of soil pH
Soils that are excessively acidic or alkaline are unsuitable for crop production. Calcium oxide (lime) is usually added to acidic soils to neutralise their acidity. When the soil is too alkaline, elemental sulfur or acidifying fertilisers like ammonium sulfate (SA) are added to remedy the condition.
Uses of salts as antacids
Antacids are substances that are in the forms of hydroxides or salts. These are used to relieve heartburn and acid build-up in the stomach. Some of the common antacids are magnesium sulfate (Epsom salt), aluminium hydroxide, and sodium hydrogencarbonate.
As inorganic fertilisers
Inorganic fertilisers are salts. Examples of these are ammonium sulfate, ammonium nitrate, and calcium phosphate.
Alleviation of health disorders
Various salts are used to alleviate problems that are associated with different health conditions. These salts include chlorides, phosphates, and sulfates of sodium, potassium, and calcium.
Production of pesticides
Certain salts are used as pesticides to kill or control pests such as insects, rodents, weeds and fungi. For example, copper(II) sulfate, iron(II) sulfate, and sodium chlorate(V) are used as pesticides for controlling fungi in crops.
Other uses of salts
There are many uses that are specific to some salts as summarised in Table below.
Table 13: Uses of some salts in daily life
 Activity 15
Activity 15
Aim: To investigate the preservative effects of table salt
Requirements: Table salt (sodium chloride-NaCl), pieces of fresh meat, beaker, petri dishes
Procedure
1. Take two pieces of fresh meat.
2. Sprinkle a generous amount of salt on one piece and leave the other without salt.
3. Leave them at room temperature for 24 hours.
4. Observe and record any appearance, texture, and odour.
Questions
1. What happened to the salted and unsalted pieces of meat?
2. What can you conclude on the effects of the salt in the two pieces of meat?
EXERCISE 4
1. Classify the following substances as neutral, acidic, or basic salts: NaCl, NaHSO₄, and Na₂CO₃. Explain your classification.
2. Describe how acidic and basic salts are formed using an example for each.
3. Differentiate between a double salt and a neutral salt, giving one example in each.
4. A salt is prepared by mixing ammonia solution and hydrochloric acids. Will the resulting salt be acidic, basic, or neutral? Explain your answer.
5. Explain the advantages of the following types of salts in real-life applications by providing two examples for each:
(a) Deliquescent salts
(b) Efflorescent salts
(c) Hygroscopic salts
PROJECT
Determination of pH values of natural and synthetic substances
Collect samples of various substances such as tap water, bottled water, river water, fruits juices, and soaps and detergents. Determine the pH values using natural and synthetic acid-base indicators, litmus papers, or pH meter.
Write a detailed report outlining the methodology, materials used, observations, results, discussions, and conclusions regarding the pH properties of the substances. Then, prepare a brief presentation summarising the experiments, results and key learnings. Enhance the presentation with visuals such as photos of the tests or a pH chart derived from the collected data.
Chapter summary
1. An acid is a chemical substance which produces hydrogen ions (H+) in water as the positively charged ions.
2. A base refers to a chemical substance which, when dissolved in water, produces hydroxide ions (OH–) as negatively charged ions. It is also defined as a metal oxide or hydroxide which neutralises an acid. An alkali is a soluble base.
3. A strong acid or strong base dissociates completely in water. A weak acid or weak base dissociates partially in water.
4. A pH indicator is a chemical substance that exhibits different colours in solutions of different acidities or alkalinities.
5. The basicity of an acid is the number of hydrogen atoms per molecule of the acid that can be displaced by a metal in a solution. An acid that has only one hydrogen atom that can be displaced is said to be monobasic. An acid with a basicity of two is dibasic, while the one with basicity of three is tribasic.
6. Neutralisation is a chemical reaction in which an acid reacts with a base to form salt and water.
7. Neutralisation is applied in various useful situations such as treating insect stings, relieving indigestion, neutralising harmful substances in the environment and manufacturing fertilisers.
8. Acids and alkalis are used in the manufacture of various industrial products.
9. Salt is an ionic compound which is made up of the positively charged ion (cation) and negatively charged ion (anion).
10. The solubility of a solute in a given solvent is the number of grams of the solute that will saturate 100 grams of a solvent at a given temperature.
11. All sodium, potassium, and ammonium salts are soluble in water.
12. All nitrates are soluble in water.
13. All chlorides are soluble in water except those of silver, mercury, and lead.
14. All sulfates are soluble in water except those of barium, calcium and lead.
15. All carbonates are insolublein water except those of sodium, potassium, and ammonium carbonates.
16. Salts have various uses. These include: controlling soil pH, as antacids, as inorganic fertilisers, alleviating health disorders, and in the preservation and seasoning of food.
Revision Exercise
Choose the correct answer for Questions 1–6. For other questions, provide the answers as per the demands indicated.
- Which of the following is a natural acid?
(a) Nitric acid
(b) Phosphoric acid
(c) Citric acid
(d) Sulfuric acid
- An acid is any substance that;
(a) contains hydrogen in its formula.
(b) dissolves in water to produce hydrogen ions.
(c) can react with NaOH.
(d) contains oxygen in its formula.
- What name is given to the process of dissolving sodium chloride in water?
(a) Precipitation process
(b) Neutralisation reaction
(c) Hydration process
(d) Dissolution process
- Which of the following salts is soluble in water?
(a) Calcium carbonate
(b) Copper carbonate
(c) Magnesium carbonate
(d) Sodium carbonate
- Which of the following substances is produced when calcium carbonate is strongly heated?
(a) Metal oxide and gas
(b) Metal oxide and smoke
(c) High heat and gas
(d) A bicarbonate and gas
- Which of the following salts decomposes to its own metal?
(a) Sodium chloride
(b) Potassium carbonate
(c) Zinc sulfate
(d) Silver nitrate
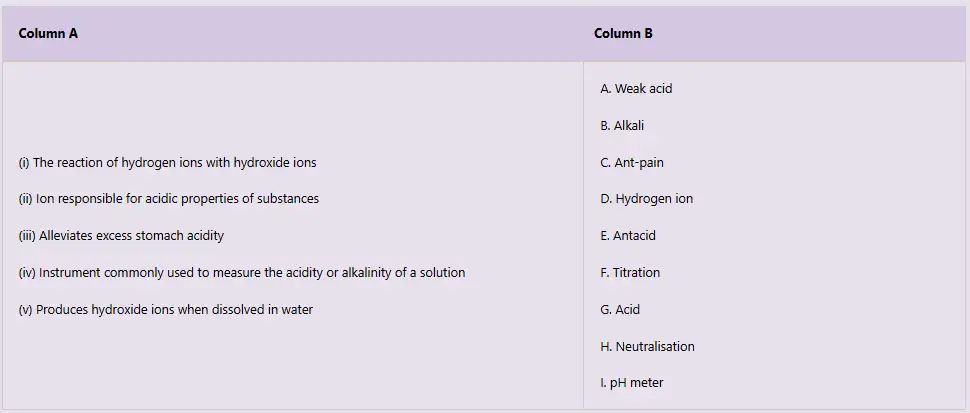
- Match the premises on properties and uses of acids and bases in Column A with their correct responses in Column B.
- Explain the following terms and illustrate their significance by providing an example for each:
(a) pH indicators
(b) Double salt
(c) Titration
(d) Solubility
- Sort out the following salts into groups of soluble and insoluble salts: Sodium carbonate, lead nitrate, lead sulfate, calcium carbonate, copper(II) sulfate, sodium nitrate, zinc chloride, silver chloride, barium carbonate, sodium sulfate, sodium chloride, potassium chloride, ammonium carbonate, and barium sulfate.
- With reasons, suggest the best methods for preparing the following compounds:
(a) Silver chloride
(b) Lead(II) sulfate
(c) Copper(II) nitrate
(d) Calcium carbonate
(e) Anhydrous zinc chloride
- What are the thermal behaviours of metals carbonates, nitrates, chlorides, and sulfates when heated? Give an example in each case.
- How are salts useful in our life in the following aspects?
(a) Food preservation and preparation
(b) Agricultural practices
(c) Medical applications
- Describe acids and bases, citing five examples in each.
- Write the balanced chemical equations and net ionic equations for the reactions between sodium hydroxide and;
(a) sulfuric acid
(b) nitric acid
- A chemistry student tested five solutions A, B, C, D, and E with a universal indicator solution to determine their pH values. The results obtained are shown in the following table:

Which solution had a;
(i) strong alkali?
(ii) strong acid?
(iii) weak acid?
(iv) neutral solution?
(v) weak base?
- During a neutralisation reaction, a student placed 25 cm3of sodium hydroxide in a flask and added few drops of phenolphthalein. The base required 22 cm³ of dilute hydrochloric acid for complete neutralisation.
(a) What apparatus should have been used to measure the exact volume of sodium hydroxide solution?
(b) What reagent was more concentrated than the other? Explain.
(c) Name the salt that was formed.
(d) How would you obtain pure crystals of the salt resulting from the solution?
(e) Write a balanced net ionic equation for the reaction.