TOPIC 1 ATOMIC STRUCTURE – CHEMISTRY FORM TWO
Concept of atomic structure
Introduction
Substances are made up of tiny particles called atoms. Atoms are the smallest particles of matter that carry an element’s properties.
In this chapter, you will learn about the concept of atomic structure and the determination of the atomic number and mass number of an element. The competencies developed will enable you to analyse the composition, behaviour, and properties of different chemical substances, enhancing your understanding of their interactions and applications in real-life situations.
Think: Atomic structure in the foundation of modern life.
Task 1
Use an interactive simulation, video, or any other reliable resource to visualise the structure of an atom and examine its sub-atomic particles.
Atoms are fundamental units that make up all matter, forming everything in the surroundings, including the air, materials, and objects used daily. Understanding atomic structure helps scientists predict the behaviour of substances and design new materials and cleaner energy.
For example, understanding the behaviour of atoms helps in food preservation, water purification, making batteries and medicines. This leads to technological and scientific developments that shape the world.
An atom is composed of smaller particles called sub-atomic particles. These include protons, neutrons, and electrons. The arrangement of these particles within an atom is referred to as the atomic structure. The structures of atoms are understood through the atomic theory, which has been developed over time through experiments and scientific discoveries.
Video on Concept of atomic structure
Task 2
Use reliable resources to analyse various atomic theory discoveries and come up with an idea about the structure of the atom.
About the year 400 BC, a Greek philosopher known as Democritus was the first to consider the idea that matter is made up of particles. Such an idea was not accepted because there was no experimental evidence to support it.
About 2000 years later, an English man called John Dalton revived the discussion. He used experimental evidence to convince people that matter is made up of particles called atoms. Through that experiment, he deduced Dalton’s spherical model of an atom shown in Figure below. Dalton’s discovery helped scientists understand chemical reactions and how substances combine.

Dalton’s model of the atom
Dalton atomic theory
In 1803, Dalton developed the theory about the atom. The four main points (assumptions) of Dalton’s Atomic Theory are summarised as follows:
1. Matter is made up of tiny particles called atoms. (The word atom means ‘unsplittable’ in Greek).
2. Atoms can neither be created nor destroyed.
3. Atoms of the same element are identical and have the same mass and properties. Atoms of a given element are different from those of any other element. Atoms of different elements can be distinguished from one another by their respective relative weights.
4. Compounds are formed by a combination of two or more different kinds of atoms. Atoms always combine in simple whole number ratios.
Dalton never imagined that anyone would ever be able to see an atom. However, modern technology has provided direct evidence that shows the positions and patterns of individual atoms. The use of modern technology has enabled scientists to carry out experiments on the atom that Dalton could not. This has led to slight modifications to Dalton’s Atomic Theory and thus formulated the so called modern concepts of Dalton’s Atomic Theory.
These modifications include the following:
1. Atoms can be either created or destroyed by means of nuclear reactions. The atom can change form through special processes such as nuclear fusion (combining the atomic nuclei) or nuclear fission (splitting the atomic nucleus). For example, an atom of uranium-235 can be split into two separate atoms.
2. Some elements have atoms of more than one kind which differ slightly in mass. Such atoms are called isotopes. For example, carbon has three isotopes known as carbon-12, carbon-13, and carbon-14.
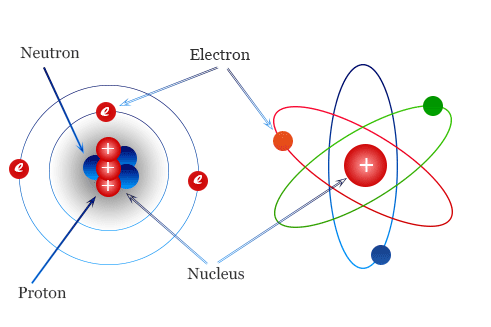
3. An atom is made up of smaller sub-atomic particles called protons, neutrons, and electrons.
4. Atoms of different elements may chemically combine in many different ratios to form compounds.
The modern atomic theory builds on Dalton’s original ideas by recognising sub- atomic particles, isotopes, and nuclear reactions while retaining the ideas of chemical combinations and reactions. Dalton’s discovery thus helped scientists understand chemical reactions and how substances combine.
Video on Dalton atomic theory
Sub-atomic particles
In 1897, J. J. Thomson carried out experiments and described an atom as a sphere of positive charge, with negative particles called electrons spread throughout the sphere. This model of the atom was referred to as plum pudding model, as shown in Figure below.

Thomson’s plum pudding model of the atom
Thomson, therefore, managed to discover the electron among the three sub- atomic particles. His discovery led to inventions of electronic devices such as televisions, radios and computers. However, another scientist called Ernest Rutherford reasoned that if Thomson’s model was correct, then the mass of the atom was evenly spread throughout the atom.
He carried out experiments and discovered that most of the mass of an atom is concentrated in the nucleus (central core) of the atom. Within the nucleus, there are positively charged particles called protons. This was the second sub-atomic particle to be discovered. Rutherford’s discovery helped scientists discover nuclear energy used to produce electricity and in radiotherapy.
Rutherford’s findings are summarised as follows:
1. Protons, the positively charged particles of an atom, are located in the nucleus.
2. Most of the mass of the atom is located in the nucleus.
3. The nucleus has a relatively smaller volume compared to the whole atom.
4. Electrons have very small masses compared to the protons.
5. Most of the space in an atom is empty.
6. Electrons are the negatively charged particles in an atom. They move around the nucleus in orbits.
Rutherford thus developed the planetary model of the atom shown in Figure below

Rutherford’s planetary model of the atom
In 1932, a scientist named James Chadwick discovered the neutrons, which also forms part of the nucleus. Figure 1.4 shows the locations of neutrons and other sub-atomic particles in an atom. Neutrons have the same mass as protons but no charge. They are located in the nucleus of an atom. They were the third sub-atomic particles to be discovered. The Chadwick discoveries have made nuclear power possible, helping to produce electricity and develop medical treatments such as cancer therapy.
The properties of neutrons are summarised as follows:
1. They have no charge (are neutral).
2. They have nearly the same mass as the corresponding protons.
3. They have a mass nearly 1840 times the mass of an electron.

Locations of sub-atomic particles in the atom
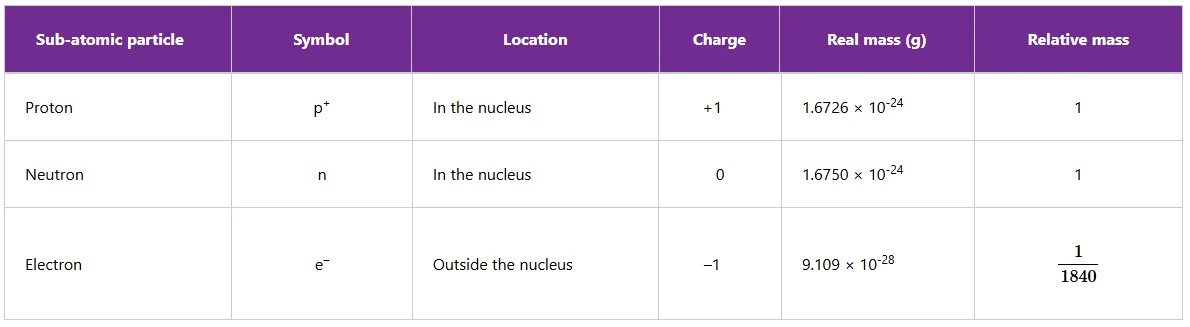
Table below summarises the properties of sub-atomic particles of an atom.
Table 1: Properties of sub-atomic particles
Activity
Aim: To build a 3D atomic model of carbon
Requirements: Six medium-sized red beads and white beads, small-sized black beads, cardboard, clay or glue and string or wire
Procedure
1. Build the nucleus by randomly arranging six red beads (protons) and six white beads (neutrons). Then, stick them together using glue to form a tight cluster at the centre.
2. Create two circular loops of varying lengths of string or wire to represent the electron shells. Attach two black beads (electrons) on the first shell, and four black beads on the second shell. Ensure they are glued and evenly spaced on the string loops.
3. Label the model and attach the entire model to a cardboard base for stability.
Questions
1. Why are the electrons arranged in different shells?
2. If one more proton is added to the nucleus of this atomic model, would it still be a carbon atom? Explain.
3. Why were different colours used for protons, neutrons and electrons in the model?
Video For Sub-atomic particles
Electronic arrangement
Task 3
Use a reliable interactive simulation to visualise the arrangements of electrons in an atom and explain how the electrons are arranged.
In 1913, Niels Bohr suggested that electrons rotate around the nucleus in special regions called shells or orbits. These shells (also known as energy levels) are at fixed distances from the nucleus. Each shell can only hold a specific number of electrons. The maximum number of electrons held within each shell can be determined by the formula 2n², where n is the position of the shell from the nucleus.
According to this formula, the:
first shell can hold (2 × 12) = 2 electrons;
second shell can hold (2 × 22) = 8 electrons; and
third shell can hold (2 × 32) = 18 electrons.
The first four shells are represented by the letters K, L, M, and N, respectively as shown in Figurebelow. Each electron in an atom is in a particular shell and the electrons must first occupy the lowest available shell nearest to the nucleus.

First four shells of an atom
For reasons beyond the scope of this book, the 3rd shell is more stable with 8 electrons.This is why even though the 3rd shell can hold up to 18 electrons, potassium element which has 19 electrons has only 8 electrons in its 3rd shell, and the last electron moves to the 4th shell. This is the same for calcium which has 20 electrons, where the last two electrons move to the 4th shell after the 3rd shell is completely filled with 8 electrons.
A shell which contains its maximum number of electrons is called a fully-filled shell. An atom with fully-filled outermost shell is said to be stable. Some atoms hold a maximum of 2 electrons (helium) or 8 electrons (neon) in their outermost shells. The elements with a maximum of 2 electrons in their outermost shells are said to exhibit a duplet state, while those with 8 electrons are said to exhibit an octet state.
Electrons are arranged so that the lowest shells are filled first. This arrangement of electrons in different shells in an atom is called electronic configuration. Figure below shows the diagrammatic electronic configurations of hydrogen, helium, neon, potassium, and sulfur atoms.

Electronic configurations of some atoms
Note that the hydrogen atom has no neutron in its nucleus. Helium and neon atoms have their outermost shells completely filled with electrons, and so they are stable atoms. The electrons are not fixed at particular positions within the shell. Instead, they move extremely fast and can be at any point within the shell. Bohr’s findings provided more information about elements that are summarised in a table called the periodic table.

Exercise 1
1. How did the discovery of the nucleus refine earlier atomic models?
2. Why are atoms considered electrically neutral under normal conditions?
3. Compare the Rutherford atomic model with the Bohr atomic model.
4. The atomic number is unique for each element in the periodic table. Explain.
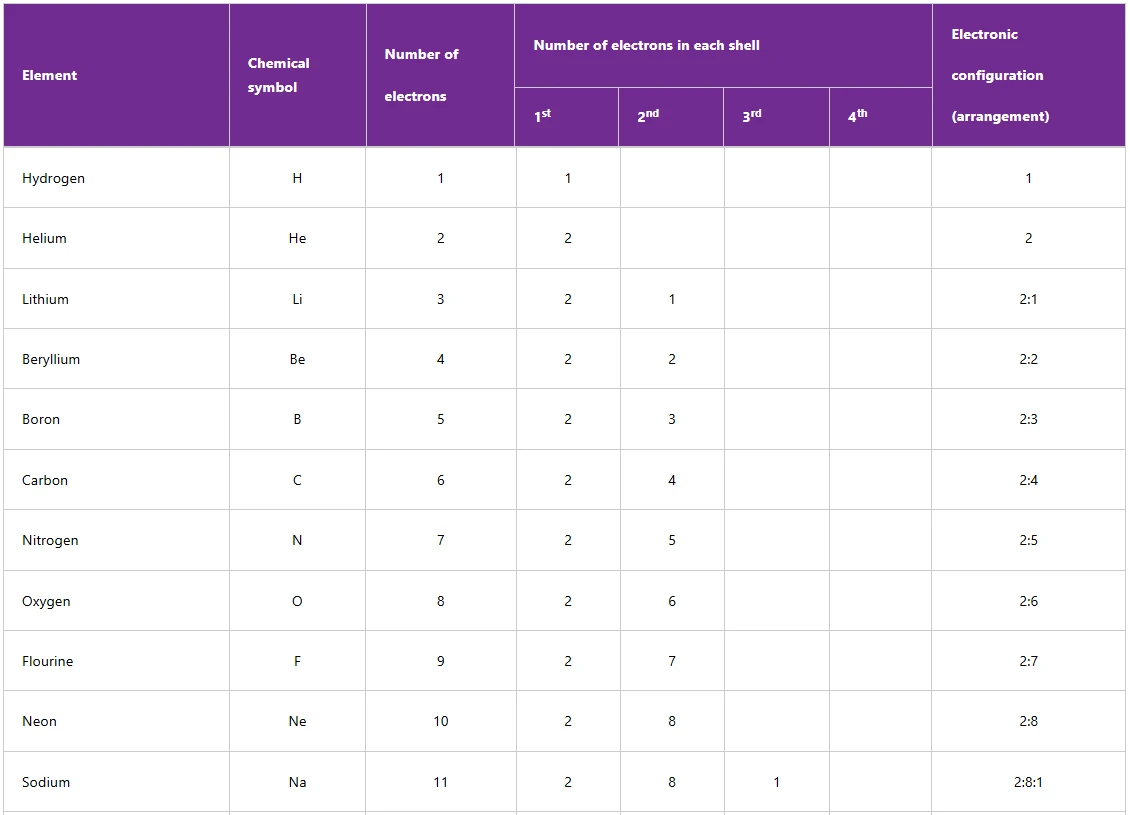
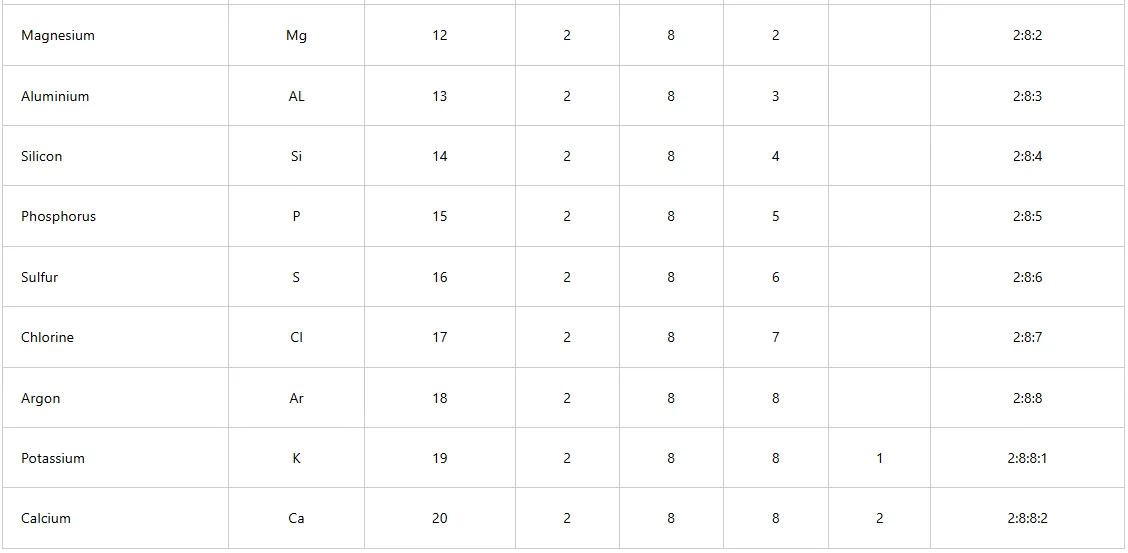
The electronic arrangements of the first 20 elements are shown in Table below
Table : Electronic arrangements of the first 20 elements
Task 4
Use drawings or cut-out pictures to represent different atomic models (Dalton’s solid sphere, Thomson’s plum pudding, Rutherford’s nucleus and Bohr’s orbits). Arrange the pictures in an order. Then, write one simple fact about each model.
Video Electronic arrangement
Determination of atomic number and mass number
Task 5
Access an online simulation on how to build an atom. Explore the simulation by adding protons, neutrons, and electrons to form different elements. Observe changes in atomic number, atomic mass, and stability of the atoms.
The sub-atomic particles of an atom, namely protons, neutrons, and electrons, relate to the atomic number and mass number of the atom.
Atomic number
The atomic number is the number of protons in an atom. It is also known as the proton number. For example, the atomic number of hydrogen is 1 since it has only one proton. A sodium atom has 11 protons in the nucleus; therefore, its atomic number is 11. Since the number of protons is equal to the number of electrons in the neutral atom, its atomic number is not only the number of protons but it is also the number of electrons.
Therefore, for the neutral atoms:
Atomic number = Number of protons = Number of electrons
Mass number
Protons and neutrons are found in the nucleus of an atom and are called nucleons. The sum of the protons and neutrons in one atom of an element is called the mass number or nucleon number or atomic mass. This number is actually taken as the mass of the atom since the mass of the electron is negligible.
Thus,
Number of protons + Number of neutrons = Mass number
For example:
(i) Hydrogen has 1 proton and 0 neutrons. Therefore, its atomic number is 1, and mass number is 1 + 0 = 1.
(ii) Boron has 5 protons and 6 neutrons. Its atomic number is 5 and mass number is 5 + 6 = 11.
(iii) Nitrogen has 7 protons and 7 neutrons. Its atomic number is 7 and mass number is 7 + 7 = 14.
It is also possible to calculate the number of neutrons and number of electrons of an atom if its mass number and atomic number are given.
Video on mass number
Chapter summary
1. An atom is the smallest particle of an element. It can only be split or destroyed by nuclear reactions.
2. There are three major sub-atomic particles, namely:
(a) protons (positively charged),
(b) neutrons (neutral), and
(c) electrons (negatively charged).
3. Protons and neutrons are located in the nucleus of an atom while electrons are found in the shells or energy levels around the nucleus.
4. The arrangement of electrons in different shells of an atom is known as electronic arrangement or electronic configuration.
5. Each shell can contain only a certain number of electrons, with the maximum being 2n², where n is the position of the shell from the nucleus.
6. For any neutral atom of an element:
Number of protons = atomic number
Number of electrons = number of protons = atomic number
Number of neutrons = mass number – atomic number
7. Isotopes are atoms of the same element with the same number of protons but different number of neutrons.
8. The relative atomic mass of an element is the average mass of one atom of the element relative to 112th the mass of one carbon-12 atom.
Revision exercise
Choose the correct answer for Questions 1−8. For other questions, provide the answers as per the demands indicated.
1. Which description corresponds to a proton?
(a) Relative mass = 1, charge = + 1
(b) Relative mass = 1, charge = 0
(c) Relative mass = 11840 , charge = –1
(d) Relative mass = 4, charge = +2.
2. What are nucleons?
(a) Neutrons and electrons
(b) Neutrons and protons
(c) Electrons and protons
(d) Protons, neutrons and electrons
3. Which atomic property is the basis for the use of carbon-14 in archaeology for dating ancient objects?
(a) Electronic configuration
(b) Radioactive decay
(c) Atomic mass
(d) Relative atomic mass
4. Most atoms are neutral because they have
(a) nucleus which is only made up of neutrons.
(b) equal numbers of electrons and protons in the shells.
(c) neutrons with zero charge.
(d) the number of electrons which balance out the number of protons in the atom.
5. Which of these statements is true about isotopes of an element?
(a) Contain the same number of protons, but different number of neutrons.
(b) The number of neutrons is the same, but the number of protons is different.
(c) Proton and neutron numbers are the same, but those of electrons are different.
(d) Electrons are added to the nucleus, with the same number of protons.
6. Which sub-atomic particles are equal in number to protons in a neutral atom?
(a) Electrons
(b) Neutrons
(c) Electrons of its ion
(d) Neutrons of its ion
7. The atomic number for an element T is 9. What is its electronic configuration?
(a) 2: 4: 3 (b) 2: 5: 2 (c) 2: 7 (d) 2: 6: 1
8. In medical diagnosis and treatment, isotopes are used in
(a) detecting malaria parasites by using special scans.
(b) using radioactive tracers for imaging and treating cancer.
(c) treating typhoid infections by using antibiotics.
(d) determining a patient’s age by checking body cells.
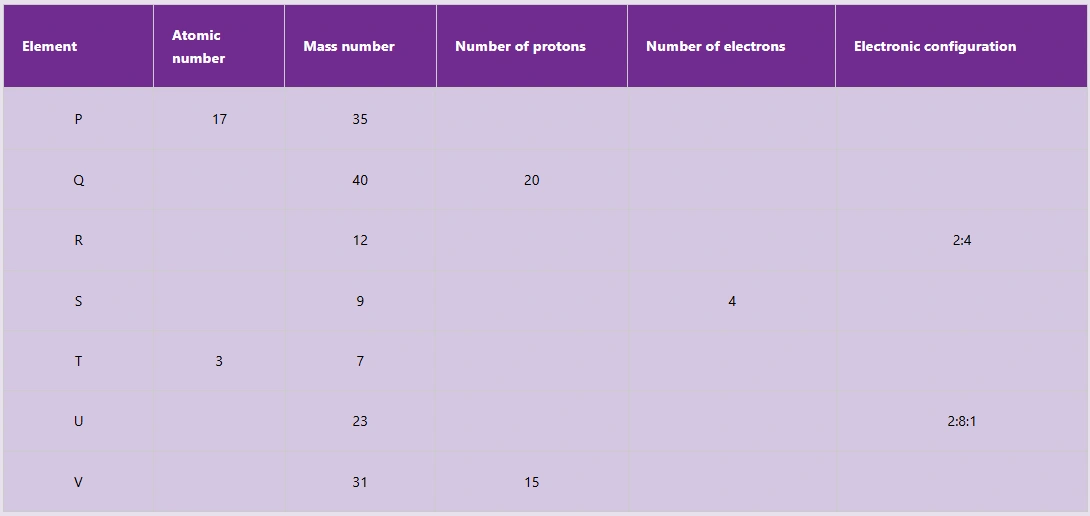
9. Copy the following table and fill the missing details of the arbitrary elements given.
10. State the number of protons, neutrons and electrons in the following atoms:
(a) 1327Al (b) 56137Ba (c) 3890Sr (d) 92235U
11. An isotope of neon has a mass number of 21 and an atomic number of 10.
(a) Write its nuclide notation.
(b) How many neutrons does it have?
(c) How many electrons does it have?
12. A sugar grain is made up of several atoms. Explain.
13. Form One students argue that atoms are visible to the naked eye because all objects are made of atoms. Explain this misconception using the atomic theory.
14. Medical doctors sometimes use X-rays to view patients’ bodies. These rays interact mainly with atoms in the patient’s bones.
(a) Which sub-atomic particle is responsible for this interaction?
(b) Why do you think this particle is important in medical imaging?
15. Describe the role of carbon-14 in determining the age of fossils.
16. Why are the relative atomic masses of elements rarely whole numbers?
17. Why is the relative atomic mass considered more practical than actual atomic mass?